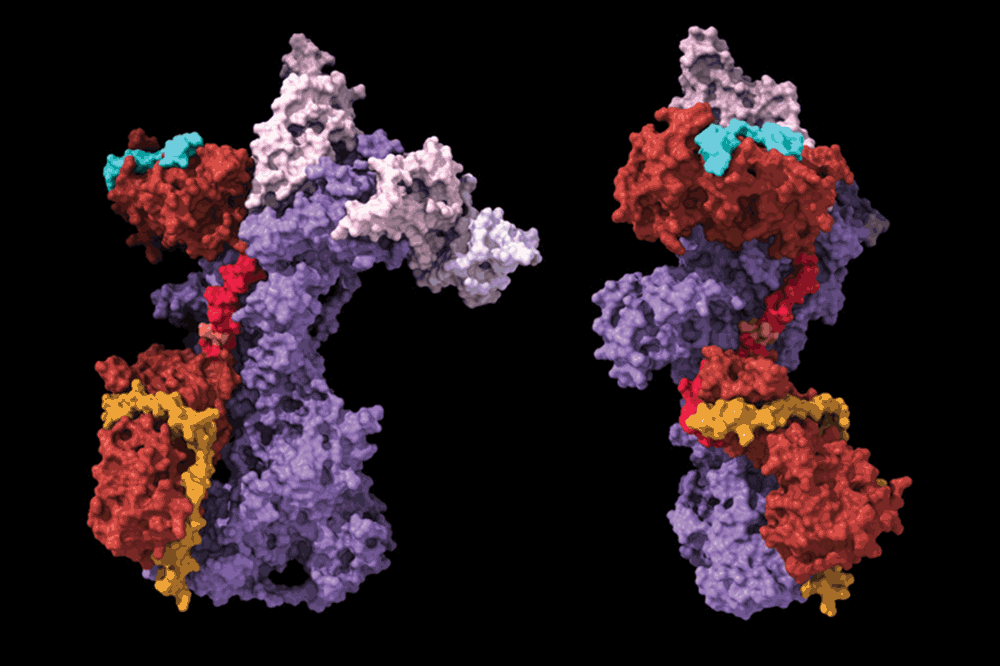Every year, over 10 million people worldwide are diagnosed with cancer. While immunotherapy has emerged as a powerful weapon in this fight, its effectiveness remains limited by our incomplete understanding of how T cells – the immune system’s targeted assassins – actually *work*. Now, groundbreaking research utilizing cryo-electron microscopy (cryo-EM) is offering an unprecedented glimpse into the structure of the T cell receptor (TCR), potentially resolving a decades-old debate and ushering in a new era of precision immunotherapy.
Decoding the T Cell Receptor: A Structural Revolution
For years, scientists have struggled to visualize the TCR in its active state. The TCR, a complex protein on the surface of T cells, is responsible for recognizing foreign invaders – antigens – and triggering an immune response. However, its small size, dynamic nature, and the challenges of isolating it in a functional state have hindered detailed structural analysis. Recent work from researchers at The Rockefeller University, published in Nature, has overcome these hurdles, providing the most detailed structural view yet of both the resting and antigen-bound states of the human TCR-CD3 complex.
This isn’t simply about filling in a structural gap. Understanding the precise conformational changes the TCR undergoes upon antigen recognition is crucial. **Cryo-EM** allows scientists to freeze biological molecules in a near-native state, then image them with electron beams, revealing their structure at near-atomic resolution. The new images reveal previously unseen details about how the TCR interacts with the CD3 complex – a crucial signaling unit – and how this interaction is modulated by antigen binding.
The Implications for Immunotherapy Design
The implications of this structural insight are far-reaching, particularly in the field of immunotherapy. Current immunotherapies, such as CAR-T cell therapy, involve genetically engineering a patient’s T cells to express a synthetic receptor that targets cancer cells. However, these therapies are often limited by off-target effects and immune suppression. A deeper understanding of the TCR’s mechanics could allow for the design of more precise and effective CAR-T cells.
Specifically, the new structural data could help researchers:
- Optimize antigen recognition: By understanding how different antigens bind to the TCR, scientists can engineer receptors with higher affinity and specificity.
- Enhance T cell signaling: The structural data reveals how the TCR interacts with the CD3 complex to initiate signaling cascades. Manipulating these interactions could boost T cell activation and function.
- Overcome immune suppression: Tumors often employ mechanisms to suppress T cell activity. Understanding the TCR’s structure could reveal new targets for overcoming these suppressive signals.
Beyond Cancer: Autoimmunity and Infectious Disease
The impact extends beyond oncology. Dysregulation of the TCR is implicated in autoimmune diseases, where the immune system mistakenly attacks the body’s own tissues. A detailed understanding of the TCR’s activation process could lead to the development of therapies that selectively dampen autoimmune responses without compromising overall immune function. Furthermore, insights into TCR structure could aid in the design of vaccines and therapies for infectious diseases, allowing for the generation of more potent and targeted immune responses.
The Rise of Computational Immunology and AI-Driven TCR Design
The wealth of structural data generated by cryo-EM is fueling a parallel revolution in computational immunology. Machine learning algorithms are now being trained to predict TCR-antigen interactions, design novel TCRs with desired specificities, and even identify potential off-target effects. This convergence of structural biology, computational modeling, and artificial intelligence promises to accelerate the development of next-generation immunotherapies.
We are entering an era where TCRs can be rationally designed, not just discovered through trial and error. This shift will dramatically reduce the time and cost associated with developing new immunotherapies, making these life-saving treatments more accessible to patients worldwide.
| Metric | Current Status (2024) | Projected Status (2030) |
|---|---|---|
| CAR-T Cell Therapy Approval Rate | ~6% | ~25% |
| Immunotherapy Market Size | $65 Billion | $200+ Billion |
| Time to Develop New TCR-Based Therapy | 8-10 Years | 3-5 Years |
Frequently Asked Questions About T Cell Receptor Research
What is the biggest challenge in developing TCR-based therapies?
The biggest challenge is predicting how a given TCR will interact with a specific antigen and avoiding off-target effects. The new structural data and computational tools are helping to address this challenge.
How will AI contribute to TCR design?
AI algorithms can analyze vast datasets of TCR-antigen interactions to identify patterns and predict the binding affinity of new TCR designs. This will significantly accelerate the development process.
When can we expect to see these advancements translate into new treatments?
While it takes time to translate research findings into clinical applications, several companies are already leveraging this new structural data to develop next-generation immunotherapies. We can expect to see early-stage clinical trials within the next 3-5 years.
The recent breakthroughs in T cell receptor structure aren’t just a scientific achievement; they represent a pivotal moment in our ability to harness the power of the immune system to fight disease. As we continue to unravel the complexities of T cell activation, the promise of personalized, highly effective immunotherapies moves ever closer to reality. What are your predictions for the future of TCR-based therapies? Share your insights in the comments below!
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.