The Silent Scourge: How Circulating Biomarkers Will Revolutionize Heterotopic Ossification Management
Nearly 30% of patients with severe trauma develop heterotopic ossification (HO)—the formation of bone in soft tissues—a debilitating condition often leading to pain, stiffness, and functional limitations. While traditionally viewed as a late-stage complication, emerging research reveals a complex interplay of circulating cells, extracellular matrix dynamics, and signaling pathways that begin *immediately* after injury. This isn’t just about treating symptoms; it’s about predicting, preventing, and ultimately, reversing aberrant bone formation using a new generation of diagnostic and therapeutic tools.
The Shifting Landscape of HO: From Reactive to Predictive
For decades, HO management has been largely reactive, relying on imaging to detect established bone and interventions like surgery or radiation to mitigate its effects. However, the limitations of these approaches—recurrence rates following excision, radiation-induced complications—highlight the need for a paradigm shift. The key lies in understanding that HO isn’t a spontaneous event, but a carefully orchestrated biological process initiated by the release of cellular and molecular signals into circulation. Heterotopic ossification is increasingly understood as a dysregulated wound healing response, and the ability to monitor this response in real-time is now within reach.
The Circulating Signature of Aberrant Bone Formation
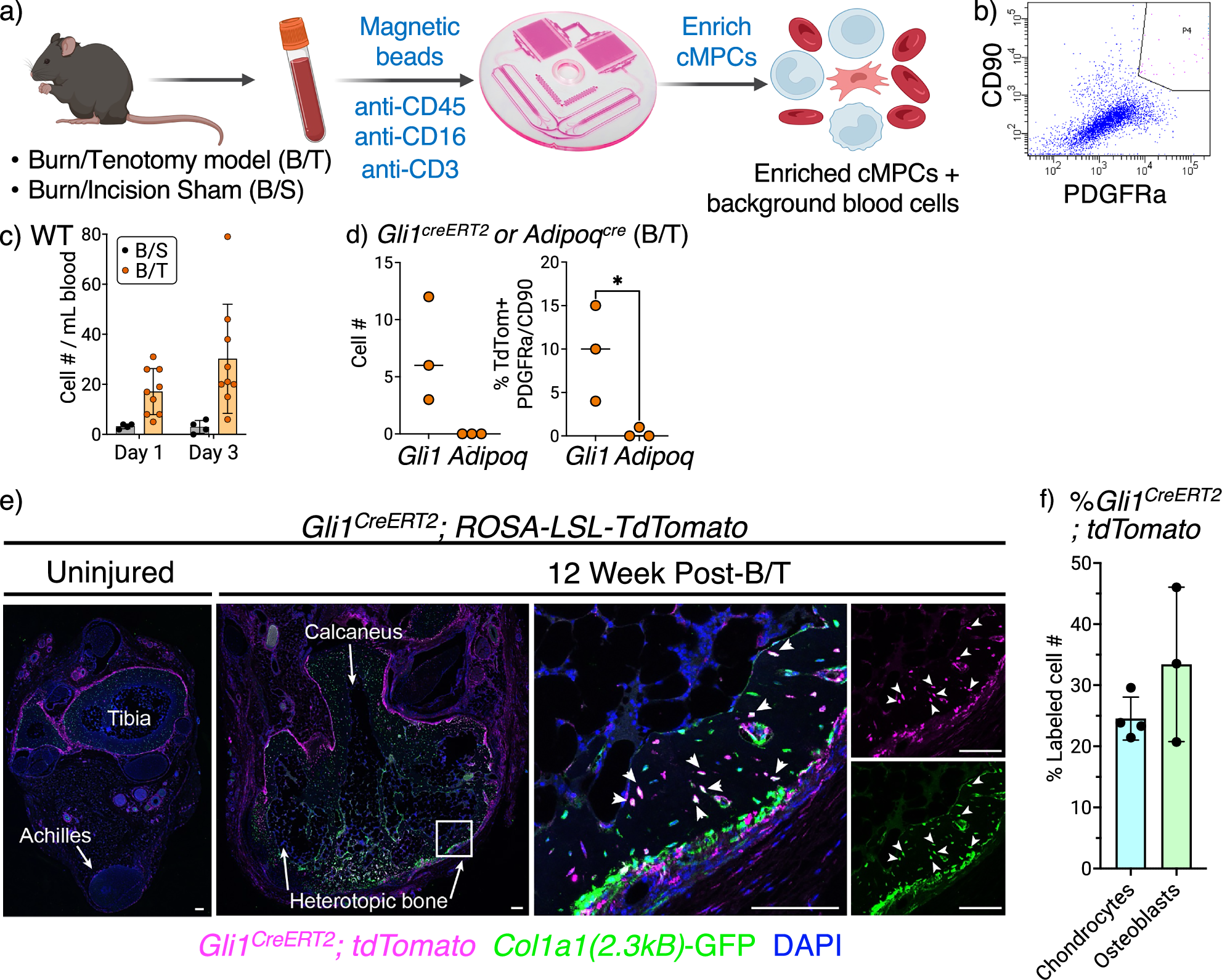
Research spearheaded by Suda et al. (2009) and further refined by Loder et al. (2018) has demonstrated the presence of circulating osteogenic precursor cells—cells capable of forming bone—in the bloodstream following trauma. These aren’t simply passive bystanders; they actively contribute to HO development. But the story is far more nuanced. Studies utilizing single-cell RNA sequencing (scRNA-seq), as pioneered by Ting et al. (2014) and Hao et al. (2024), are revealing that these circulating cells exhibit distinct gene expression profiles depending on the type and severity of injury. Furthermore, the work of Sullivan et al. (2014) and others demonstrates that even cells originating from distant sites, like brain tumors, can adopt mesenchymal characteristics and contribute to ectopic tissue formation.
Beyond Osteogenic Precursors: The Role of Monocytes and the Extracellular Matrix
It’s not just about the cells *capable* of forming bone, but the environment that guides their fate. Sorkin et al. (2020) showed that monocytes play a crucial regulatory role in HO, influencing the inflammatory response and subsequent bone formation. Simultaneously, the extracellular matrix (ECM) undergoes dramatic remodeling after injury, as highlighted by Huber et al. (2020). The interplay between these factors—circulating cells, immune response, and ECM composition—is critical. Recent work by Kang et al. (2024) emphasizes the role of the HIF-1α/PLOD2 axis in integrating ECM organization and cell metabolism, leading to aberrant musculoskeletal repair. Understanding these interactions is paramount.
Emerging Diagnostic Technologies: A Glimpse into the Future
The ability to detect these early molecular signatures opens the door to non-invasive diagnostic tools. While traditional radiographic imaging (Brownley et al., 2015) often lags behind the initial biological events, newer technologies are showing promise. Peterson et al. (2013) demonstrated the potential of transcutaneous Raman spectroscopy for early detection in burn patients. Perosky et al. (2014) expanded on this with near-infrared optical imaging, revealing dynamic turnover and progression of mineralization. However, the most significant advancements are likely to come from liquid biopsies—analyzing circulating biomarkers in blood samples. The techniques developed for circulating tumor cell (CTC) detection, such as those by Mishra et al. (2020) and Karabacak et al. (2014), are directly applicable to HO, allowing for the identification and quantification of osteogenic precursors and inflammatory markers.
Therapeutic Horizons: Targeting the Root Cause
Current treatments for HO are often palliative. However, a deeper understanding of the underlying mechanisms is paving the way for targeted therapies. The re-emergence of mesenchymal stem cell populations after surgical excision, as shown by Agarwal et al. (2017), underscores the need for strategies that address the root cause of aberrant bone formation. Research into signaling pathways like Hedgehog (Kan et al., 2018) and NGF-TrkA (Lee et al., 2021) offers potential therapeutic targets. Furthermore, manipulating the ECM—perhaps through the use of inhibitors of lysyl oxidases (Vallet & Ricard-Blum, 2019) or PCPE-2 (Vadon-Le Goff et al., 2023)—could disrupt the process of mineralization. The discovery of Vegfc-expressing cells forming heterotopic bone (Vishlaghi et al., 2024) also presents a novel avenue for intervention.
The Promise of Personalized Medicine in HO Management
The future of HO management isn’t about a one-size-fits-all approach. It’s about personalized medicine, tailored to the individual patient’s risk factors, injury profile, and circulating biomarker signature. By combining advanced diagnostic tools with targeted therapies, we can move beyond simply treating the symptoms of HO and towards preventing its occurrence altogether. The convergence of single-cell technologies, liquid biopsies, and a deeper understanding of the cellular and molecular mechanisms driving aberrant bone formation promises a new era in the management of this often-overlooked, yet profoundly debilitating, condition.
Frequently Asked Questions About Heterotopic Ossification
What is the long-term outlook for patients with HO?
The long-term outlook varies significantly depending on the location and extent of the HO. Untreated HO can lead to chronic pain, limited range of motion, and functional disability. However, with early detection and appropriate management, many patients can achieve significant improvements in their quality of life.
<h3>Are there any lifestyle modifications that can help prevent HO?</h3>
<p>While there’s no guaranteed way to prevent HO, maintaining good overall health, avoiding prolonged immobilization, and following a physician’s recommendations for post-injury care can help reduce the risk. Further research is needed to identify specific dietary or exercise interventions that may be beneficial.</p>
<h3>How will circulating biomarkers change the way HO is treated?</h3>
<p>Circulating biomarkers will allow for earlier detection of HO risk, enabling proactive interventions before significant bone formation occurs. They will also help identify patients who are most likely to benefit from specific therapies, leading to more personalized and effective treatment strategies.</p>The era of reactive HO management is drawing to a close. As our understanding of the underlying biological processes deepens, and as new diagnostic and therapeutic tools become available, we are poised to transform the lives of patients affected by this silent scourge. What are your predictions for the future of HO diagnostics and treatment? Share your insights in the comments below!
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.