FDA Clears Eyas Medical Imaging’s Novel MRI System for Neonates
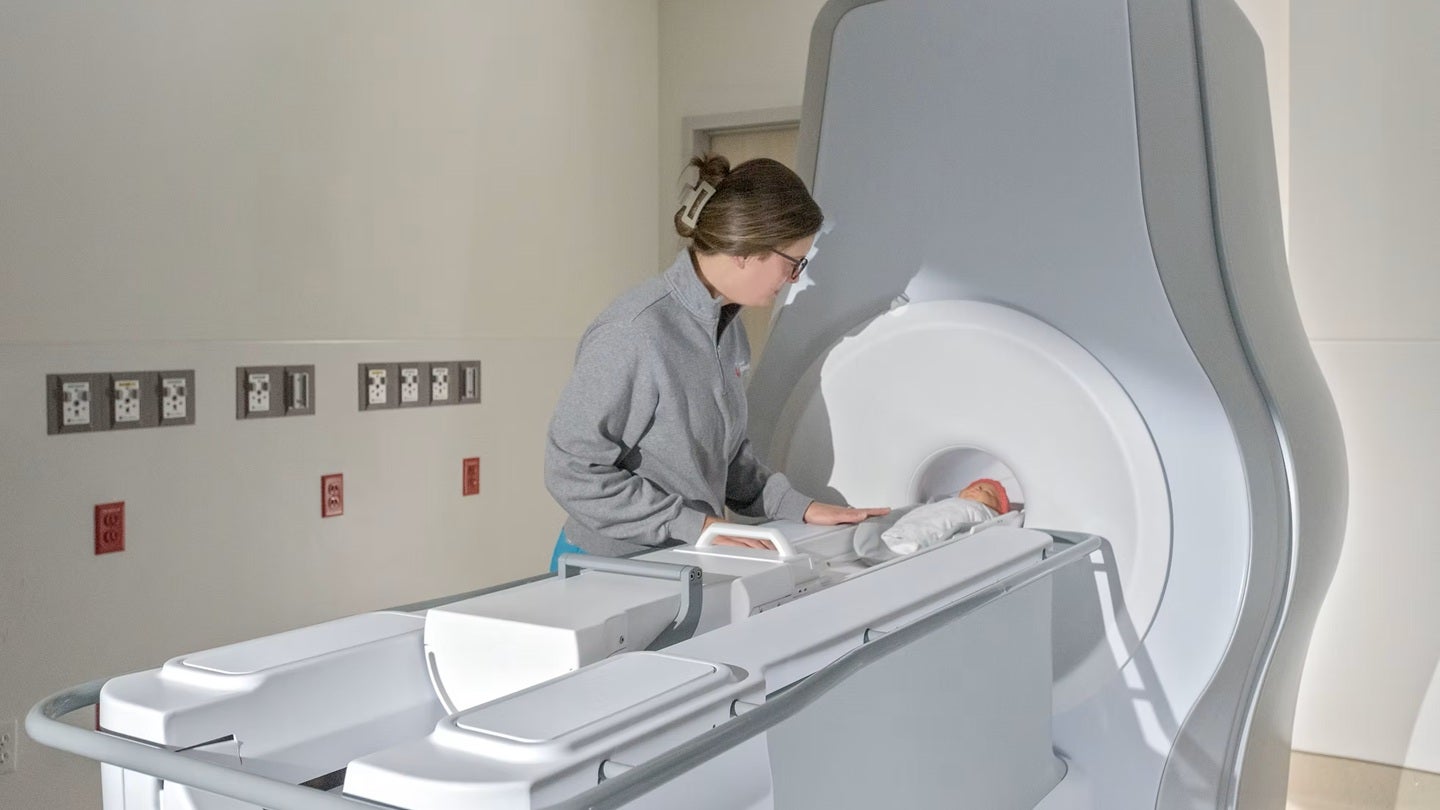
In a significant advancement for pediatric medical imaging, the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to Eyas Medical Imaging for its Ascent3T neonatal MRI system. This innovative device is specifically designed to address the unique challenges of imaging newborns and infants, promising earlier and more accurate diagnoses of critical conditions.
The Ascent3T system represents a departure from traditional MRI technology, which often requires lengthy scan times and can be difficult for the smallest patients to tolerate. Eyas Medical Imaging engineered the Ascent3T with a focus on minimizing scan duration and maximizing patient comfort, crucial factors when dealing with vulnerable neonates. The system’s design takes into account the smaller anatomy and physiological differences of infants, leading to improved image quality and diagnostic confidence.
The Critical Need for Specialized Neonatal Imaging
Neonatal MRI is a vital tool in the diagnosis of a wide range of conditions, including brain injuries, congenital heart defects, and metabolic disorders. Early detection of these conditions is paramount, as timely intervention can significantly improve outcomes. However, conventional MRI systems are not always ideal for neonates. The challenges include motion artifacts, the need for sedation, and the difficulty of positioning infants comfortably within the scanner.
The Ascent3T system aims to overcome these hurdles. Its advanced gradient coils and optimized radiofrequency design enable faster imaging speeds, reducing the need for sedation and minimizing patient discomfort. Furthermore, the system’s compact size and flexible coil configurations allow for easier positioning of infants, improving image quality and diagnostic accuracy. What impact will this have on the future of neonatal care? Could this technology eventually be adapted for use in premature infants, further expanding its diagnostic capabilities?
The 510(k) clearance pathway, used by the FDA for medical devices substantially equivalent to those already legally marketed, signifies that the Ascent3T system meets the agency’s standards for safety and effectiveness. This clearance allows Eyas Medical Imaging to begin commercializing the system, bringing this potentially life-saving technology to hospitals and neonatal intensive care units (NICUs) across the United States.
Beyond the technical specifications, the development of the Ascent3T highlights a growing trend in medical device innovation: the focus on patient-specific solutions. As healthcare providers increasingly recognize the importance of tailoring treatments to individual needs, we can expect to see more devices designed with specific patient populations in mind. The FDA’s role in evaluating and clearing these devices is crucial to ensuring their safety and efficacy.
Eyas Medical Imaging’s commitment to neonatal imaging extends beyond the Ascent3T system. The company is actively involved in research and development, exploring new technologies and techniques to further improve the diagnosis and treatment of neonatal conditions. RSNA News provides further details on the system’s capabilities.
Frequently Asked Questions About the Ascent3T Neonatal MRI System
-
What is a neonatal MRI and why is it important?
A neonatal MRI is a specialized imaging technique used to visualize the internal structures of newborns and infants. It’s crucial for diagnosing conditions that may not be apparent through other methods, enabling early intervention and improved outcomes.
-
How does the Ascent3T system differ from traditional MRI machines?
The Ascent3T is specifically engineered for the anatomy and physiology of neonates, featuring faster scan times, reduced need for sedation, and optimized image quality for smaller patients.
-
What conditions can the Ascent3T help diagnose in newborns?
The system can aid in the diagnosis of a wide range of conditions, including brain injuries, congenital heart defects, metabolic disorders, and other developmental abnormalities.
-
What does FDA 510(k) clearance mean for the Ascent3T system?
FDA 510(k) clearance indicates that the Ascent3T is substantially equivalent to a legally marketed device and meets the agency’s standards for safety and effectiveness, allowing it to be commercially available.
-
Will the Ascent3T system require changes to current hospital workflows?
While some training may be required for staff, the Ascent3T is designed to integrate seamlessly into existing hospital imaging departments, minimizing disruption to current workflows.
-
What is the future outlook for neonatal MRI technology?
The future of neonatal MRI is bright, with ongoing research focused on further improving image quality, reducing scan times, and expanding the range of conditions that can be diagnosed.
The FDA’s clearance of the Ascent3T system marks a pivotal moment in neonatal medical imaging. This technology has the potential to transform the way we diagnose and treat infants, offering hope for improved outcomes and a brighter future for vulnerable newborns.
Share this groundbreaking news with your network and join the conversation below. What are your thoughts on the potential impact of this technology on neonatal care?
Disclaimer: This article provides general information and should not be considered medical advice. Always consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.