The fight against Alzheimer’s disease is entering a new phase, moving beyond simply slowing cognitive decline to potentially *preventing* plaque formation and reversing existing damage. While recently approved monoclonal antibody treatments offer a modest extension of independent living – roughly 10 months – a groundbreaking new immunotherapy developed at Washington University School of Medicine in St. Louis offers a potentially more impactful, and significantly more convenient, approach. This research, published in Science, leverages the brain’s own support cells, astrocytes, to clear the amyloid plaques long believed to be a primary driver of the disease. This isn’t just incremental progress; it’s a fundamentally different strategy with the potential to reshape Alzheimer’s treatment.
- Single Injection Potential: Unlike current antibody therapies requiring frequent infusions, this CAR-astrocyte therapy demonstrated efficacy in mice with a single injection.
- Dual Action: The therapy both prevented plaque formation in early-stage models *and* reduced existing plaque levels by 50% in advanced stages.
- Beyond Alzheimer’s: Researchers believe this “reprogramming” approach could be adapted to treat other neurodegenerative diseases and even brain tumors.
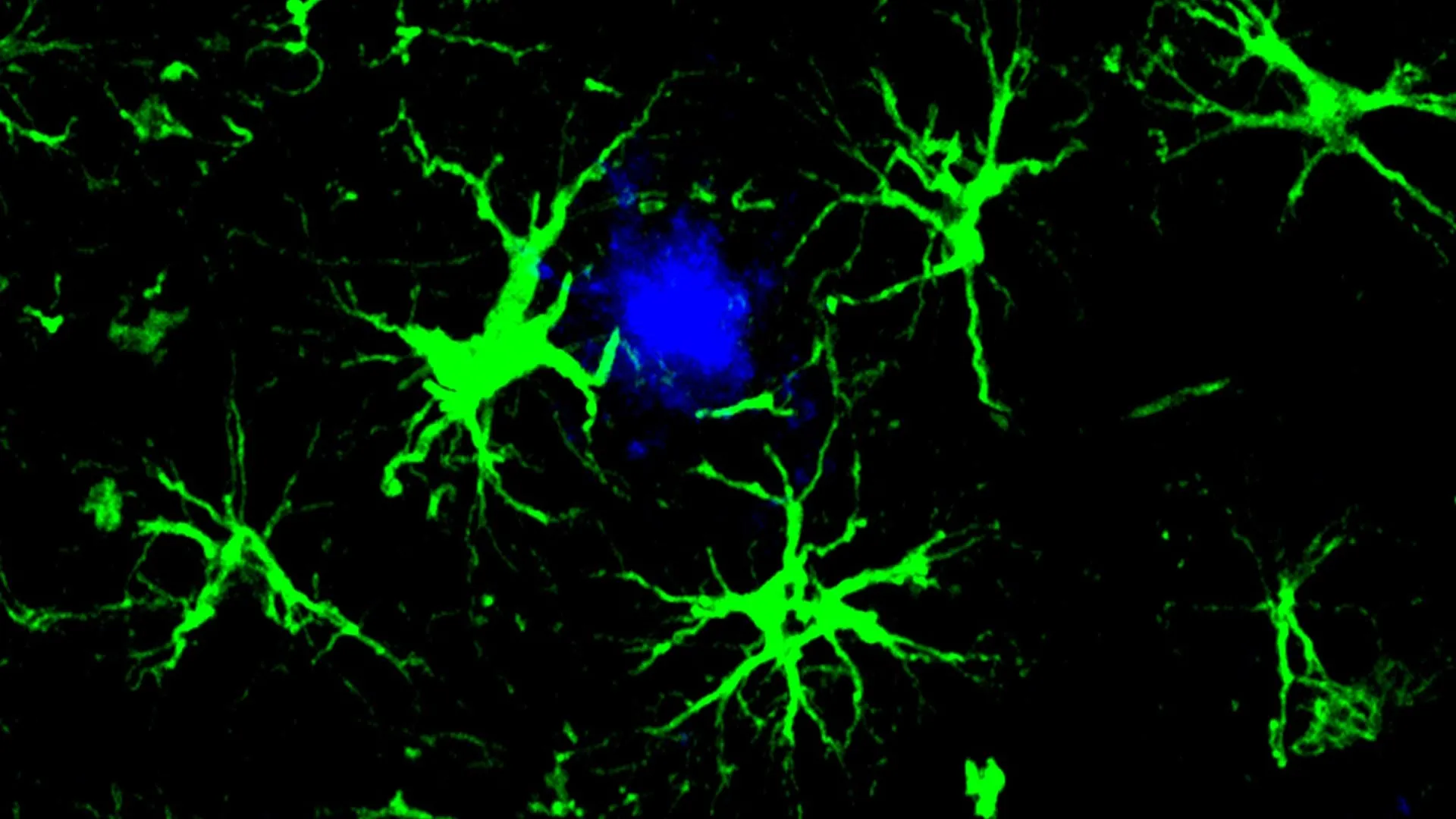
For decades, the prevailing theory in Alzheimer’s research has centered on the accumulation of amyloid beta plaques and neurofibrillary tangles. While the amyloid hypothesis has faced some challenges, the recent FDA approvals of lecanemab (Leqembi) and aducanumab (Aduhelm) – both monoclonal antibodies targeting amyloid – validate the importance of addressing these protein deposits. However, these drugs are expensive, carry risks of side effects like brain swelling, and require burdensome administration schedules. The brain’s natural cleanup crew, microglia, often become overwhelmed in Alzheimer’s, unable to effectively clear the accumulating amyloid. This new research circumvents that limitation by enlisting a different type of brain cell – astrocytes – and equipping them with a targeted plaque-removal capability.
The team, led by Marco Colonna and Yun Chen, drew inspiration from the success of CAR-T cell therapy in cancer. CAR-T therapy genetically engineers a patient’s own T cells to recognize and destroy cancer cells. Here, the researchers engineered astrocytes, the most abundant cells in the brain responsible for maintaining a healthy environment for neurons, to express a chimeric antigen receptor (CAR). This CAR acts like a homing device, allowing the astrocytes to specifically bind to and engulf amyloid beta plaques. The results in mice are compelling: complete prevention of plaque formation in early-stage models and a significant reduction in existing plaques in later-stage models. This suggests a potential therapeutic window, but also hints at the possibility of proactive intervention before symptoms even manifest.
The Forward Look
The most immediate next step is, of course, rigorous safety testing and refinement of the therapy in larger animal models. The researchers have already filed a patent, signaling their intent to move this technology towards clinical trials. However, several hurdles remain. Ensuring the engineered astrocytes don’t disrupt normal brain function is paramount. Precise targeting is crucial to avoid unintended consequences. Furthermore, translating mouse studies to human efficacy is notoriously difficult.
Despite these challenges, the potential impact is enormous. A single-injection therapy that could halt or reverse Alzheimer’s progression would be a game-changer. Beyond Alzheimer’s, the adaptability of this CAR-astrocyte approach is particularly exciting. The ability to reprogram astrocytes to target different markers opens doors to treating a wide range of neurological disorders, including brain tumors. We can anticipate a surge in research exploring this “cellular immunotherapy” platform, potentially ushering in a new era of precision medicine for the brain. The next 2-3 years will be critical as this promising therapy moves closer to human trials, and the scientific community closely watches for signs of both efficacy and safety.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.