The subtle differences in our genetic code, those seemingly insignificant variations, are increasingly revealing themselves as pivotal in explaining the divergence between humans and our closest primate relatives. New research published in Science Advances highlights one such difference – a silenced gene in chimpanzees, LINC00662, crucial for neuron development in humans – and demonstrates the power of CRISPR technology to unlock evolutionary secrets. This isn’t just about understanding our past; it’s about potentially illuminating the genetic basis of uniquely human neurological conditions.
- Gene Silencing & Evolution: A retroviral insertion silences LINC00662 in chimpanzees, hindering neuron development compared to humans.
- CRISPR’s Role: CRISPR-Cas9 successfully reactivated the gene in chimpanzee cells, demonstrating a direct link between the insertion and gene expression.
- Non-Coding DNA Matters: The study underscores the importance of investigating non-coding regions of the genome, often dismissed as “junk DNA,” for evolutionary insights.
For years, scientists have known that humans and chimpanzees share approximately 98% of their DNA. However, the 2% difference accounts for profound distinctions in cognitive abilities and susceptibility to disease. This study focuses on a specific instance where that 2% makes a significant impact. The LINC00662 gene, when active in humans, plays a critical role in axon extension and dendrite formation – the very building blocks of neural networks. The research team discovered that this gene is effectively “switched off” in chimpanzees due to an insertion of a retrovirus, PTERV1, into its sequence. Retroviral insertions are common occurrences in genomes, often silenced by the host or, occasionally, co-opted for new functions (as seen with the ARC gene involved in memory). The fact that PTERV1 silenced LINC00662 in the chimpanzee lineage is particularly intriguing.
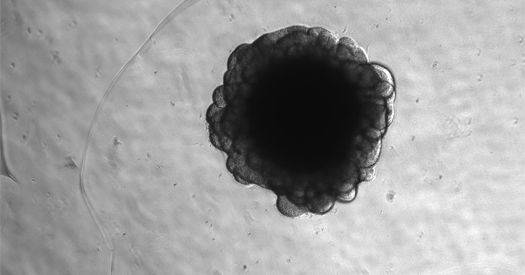
The ability to use CRISPR-Cas9 to excise the PTERV1 insertion and reactivate LINC00662 in chimpanzee induced pluripotent stem cells is a technical breakthrough. It wasn’t possible until recent advances in long-read sequencing and, crucially, the development of chimpanzee brain organoids – miniature, 3D models of the brain grown in a lab. These organoids, while still requiring refinement, provide a crucial platform for studying human-specific genetic functions in a more relevant context than traditional cell cultures.
The Forward Look
This research opens several exciting avenues. First, understanding *exactly* what LINC00662 does in developing human neural tissue is paramount. Researchers are already exploring “humanized” mouse models to study the gene’s function. More importantly, this work highlights the potential for identifying other “human-specific” genes – genes active in humans but silenced in other primates – that contribute to our unique cognitive abilities. Could variations in these genes also be linked to neurological disorders like autism or schizophrenia? The study’s authors suggest this is a strong possibility. Furthermore, the investigation into PTERV1 and other endogenous retroviruses could reveal broader mechanisms by which these viral elements regulate gene expression and influence brain evolution across primate species. Expect to see increased investment in organoid technology and long-read sequencing as researchers race to decode the genetic basis of what makes us human. The focus will likely shift towards identifying other retroviral insertions and their impact on gene regulation in primates, potentially revealing a hidden layer of complexity in our evolutionary history.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.