Beyond Carbon Capture: A New Catalyst Poised to Revolutionize Sustainable Fuel Production
Every year, over 50 billion tons of carbon dioxide are released into the atmosphere. While carbon capture technologies are gaining traction, simply *storing* CO₂ isn’t a long-term solution. What if we could transform this greenhouse gas into something valuable? Researchers at the Chinese Academy of Sciences have achieved a breakthrough, developing a novel catalyst that converts CO₂ into methanol with unprecedented efficiency – a threefold increase at 300°C. This isn’t just an incremental improvement; it’s a potential paradigm shift in sustainable fuel production, and a crucial step towards a circular carbon economy.
The Challenge of CO₂ Conversion: Why Spatial Decoupling Matters
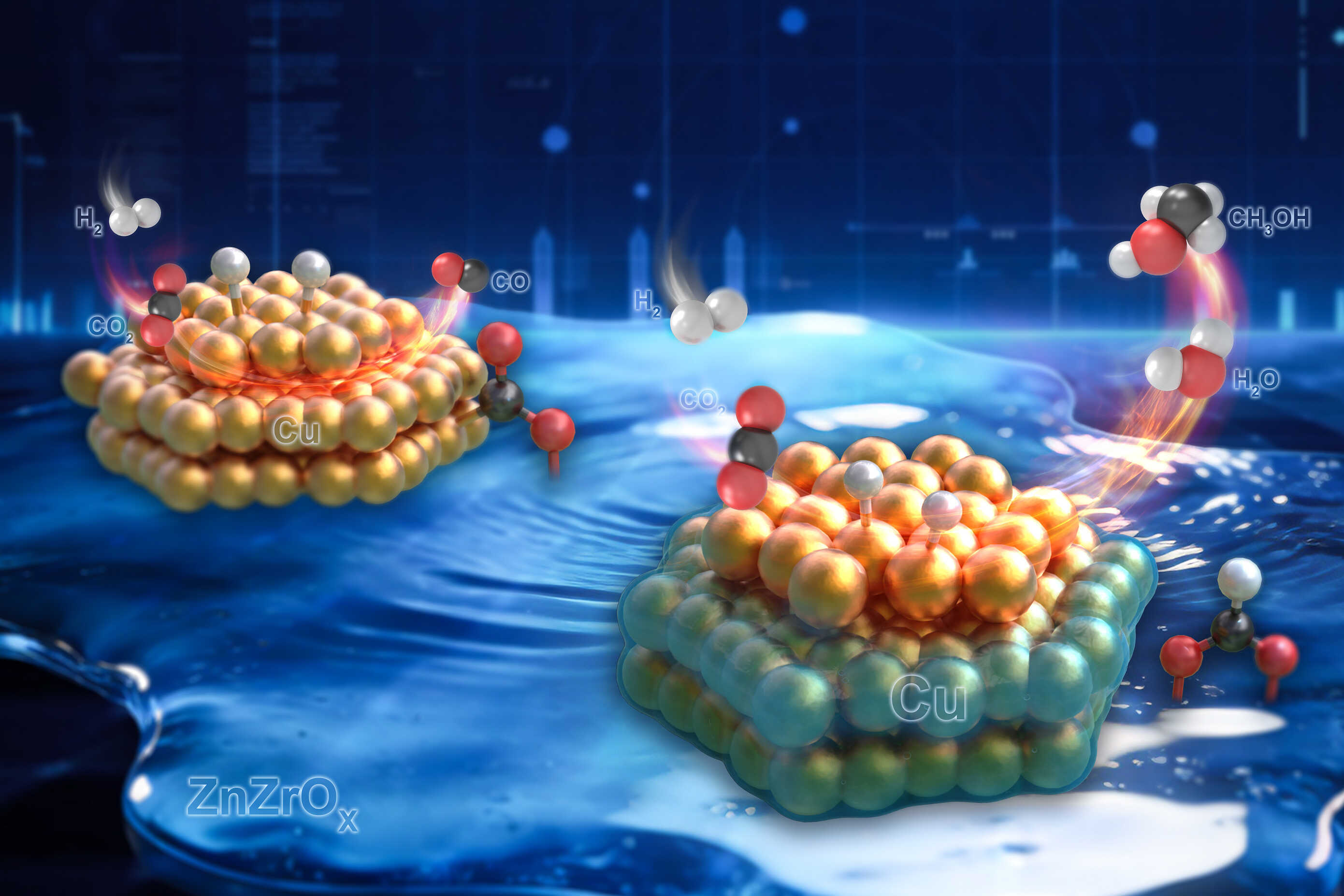
Converting carbon dioxide into usable fuels like methanol is notoriously difficult. The process requires breaking the incredibly stable carbon-oxygen bond, demanding significant energy input and highly efficient catalysts. Traditional catalysts often suffer from limitations – active sites becoming saturated or poisoned, leading to reduced performance and selectivity. The key innovation lies in a strategy called spatially decoupling active sites.
This approach, as detailed in recent publications from Phys.org and SciTechDaily, doesn’t rely on a single, multifunctional catalyst. Instead, it separates the crucial steps of CO₂ adsorption and methanol synthesis onto distinct, spatially separated sites. This prevents interference and allows each site to operate at peak efficiency. Think of it like an assembly line – each station focuses on a specific task, maximizing overall output.
How the New Catalyst Works: A Two-Step Process
The Chinese Academy of Sciences team engineered a catalyst featuring isolated copper atoms for CO₂ adsorption and a separate site containing indium nanoparticles for methanol synthesis. This separation is achieved through a carefully designed support material. The isolated copper atoms effectively ‘capture’ the CO₂ molecules, bringing them into close proximity with the indium nanoparticles, which then facilitate the conversion into methanol. This precise control over the reaction environment is what drives the dramatic improvement in yield.
The Methanol Economy: Beyond Fuel, Towards a Sustainable Chemical Feedstock
Methanol (CH₃OH) is often touted as a potential alternative fuel, but its applications extend far beyond transportation. It’s a versatile chemical feedstock used in the production of plastics, adhesives, solvents, and even pharmaceuticals. A readily available, sustainably produced source of methanol could significantly reduce our reliance on fossil fuels across multiple industries.
The implications are particularly profound for regions with limited access to traditional energy sources. Decentralized methanol production, utilizing locally sourced CO₂ emissions, could empower communities and foster energy independence. Furthermore, methanol can be easily transported and stored, making it an ideal energy carrier.
| Metric | Traditional Catalysts | New Spatially Decoupled Catalyst |
|---|---|---|
| CO₂ to Methanol Yield | Baseline | 3x Baseline at 300°C |
| Reaction Temperature | Higher | 300°C (Lower Energy Input) |
| Catalyst Stability | Lower | Improved (Reduced Site Saturation) |
Scaling Up and Future Directions: The Road to Commercialization
While the results are incredibly promising, several challenges remain before this technology can be deployed on a large scale. Scaling up the production of the catalyst itself is a key hurdle. Ensuring the long-term stability and durability of the catalyst under industrial conditions is also crucial. Further research will focus on optimizing the support material and exploring alternative metal combinations to enhance performance and reduce costs.
However, the momentum is building. We’re likely to see increased investment in CO₂ conversion technologies, driven by both environmental concerns and the economic potential of a circular carbon economy. The development of more efficient catalysts, coupled with advancements in carbon capture and utilization (CCU) technologies, will be essential for achieving a sustainable future.
Frequently Asked Questions About CO₂ to Methanol Conversion
What is the biggest advantage of this new catalyst?
The primary advantage is the threefold increase in methanol yield at a relatively low temperature (300°C) compared to traditional catalysts. This significantly improves the efficiency and reduces the energy requirements of the CO₂ conversion process.
How does spatially decoupling active sites improve catalyst performance?
By separating the CO₂ adsorption and methanol synthesis steps onto distinct sites, the catalyst prevents interference between these processes. This allows each site to operate at its optimal efficiency, leading to higher overall performance.
What are the potential applications of sustainably produced methanol?
Methanol can be used as a fuel, a chemical feedstock for producing plastics and other materials, and as a solvent. Sustainable methanol production could reduce our reliance on fossil fuels across multiple industries.
What are the next steps in developing this technology?
The next steps involve scaling up catalyst production, improving its long-term stability, and optimizing the process for industrial applications. Further research will also explore alternative metal combinations and support materials.
The breakthrough from the Chinese Academy of Sciences isn’t just about a new catalyst; it’s about reimagining our relationship with carbon dioxide. It’s a powerful demonstration of how innovative materials science can pave the way for a more sustainable and circular future, transforming a major pollutant into a valuable resource. The race is now on to translate this laboratory success into a global-scale solution.
What are your predictions for the future of CO₂ conversion technologies? Share your insights in the comments below!
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.