FDA Greenlights Wave of AI-Powered Medical Innovations, Transforming Patient Care
The U.S. Food and Drug Administration (FDA) has recently authorized a significant number of cutting-edge medical technologies, signaling a pivotal shift towards artificial intelligence (AI) and advanced diagnostics in healthcare. These clearances promise to enhance precision, reduce invasiveness, and broaden access to critical medical services.
The Rise of AI in Modern Medicine
The latest FDA approvals underscore a growing trend: the integration of AI and sophisticated technology into clinical workflows. This isn’t simply about automating existing processes; it’s about fundamentally changing how diseases are detected, managed, and ultimately, treated. From wearable sensors providing continuous physiological data to AI algorithms analyzing complex medical images, the potential for improved patient outcomes is substantial.
Wearable Technology and Remote Patient Monitoring
A key area of innovation lies in wearable devices capable of continuous, long-term monitoring. Carré Technologies Inc. (dba Hexoskin) has received 510(k) clearance for its Hexoskin Medical System (HMS), a smart biometric shirt that delivers continuous ECG, respiratory monitoring, and activity tracking for ambulatory patients. This breakthrough allows for remote assessment of arrhythmias, such as atrial fibrillation, and detailed analysis of breathing patterns outside of traditional clinical settings.
The HMS isn’t just for direct patient care. It’s poised to revolutionize clinical research, enabling decentralized trials and the collection of high-resolution, real-world data for the development of AI-driven digital biomarkers across a range of specialties, including cardiology, pulmonology, and neurology.
AI-Enhanced Cardiac and Aortic Imaging
AI is also making significant strides in cardiovascular imaging. RapidAI’s Aortic Management solution, recently cleared by the FDA, utilizes deep clinical AI to transform the assessment and management of aortic disease. The system automatically generates precise measurements, creates 3D reconstructions, and tracks anatomical changes over time, aiding in the identification and monitoring of aortic pathology.
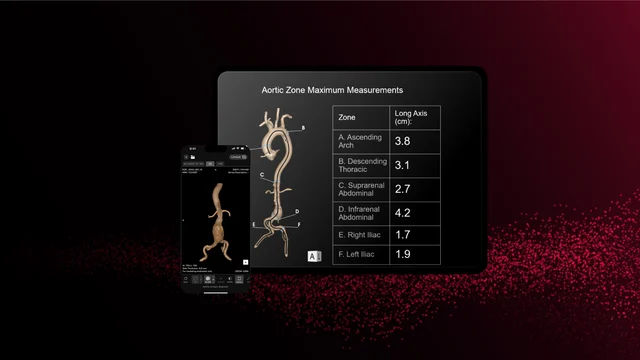
Unlike basic triage tools, Rapid Aortic supports the entire patient journey – from initial screening to long-term surveillance. Its ability to process both contrast and non-contrast CT scans expands its applicability across various care settings. Clinicians benefit from reduced cognitive load, faster read times, and improved accuracy, while surgeons gain access to precise visualizations for pre-operative planning.
Circle Cardiovascular Imaging Inc. (Circle CVI) has also secured FDA clearance for its cvi42 | Plaque solution, an AI-powered technology for comprehensive coronary plaque analysis. This on-premise system quantifies plaque buildup, aiding in precise risk stratification. Notably, this clearance coincides with the implementation of a new Category I CPT code (75XX6) in January 2026, solidifying plaque quantification as a standard clinical practice.
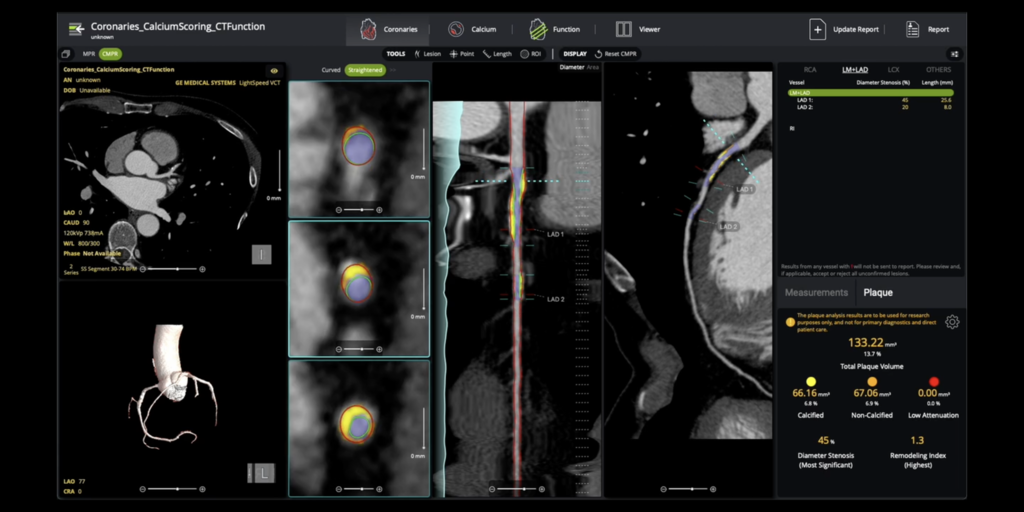
Further advancing cardiac diagnostics, Bunkerhill Health achieved a first-of-its-kind FDA clearance for its Bunkerhill MAC algorithm, designed to detect and quantify mitral annular calcification (MAC) on routine chest CT scans. This often-overlooked finding is linked to increased cardiovascular risk, and the AI-powered tool integrates into Bunkerhill’s Carebricks platform to support informed clinical decisions.

Expanding Horizons: Neuroscience and Pain Management
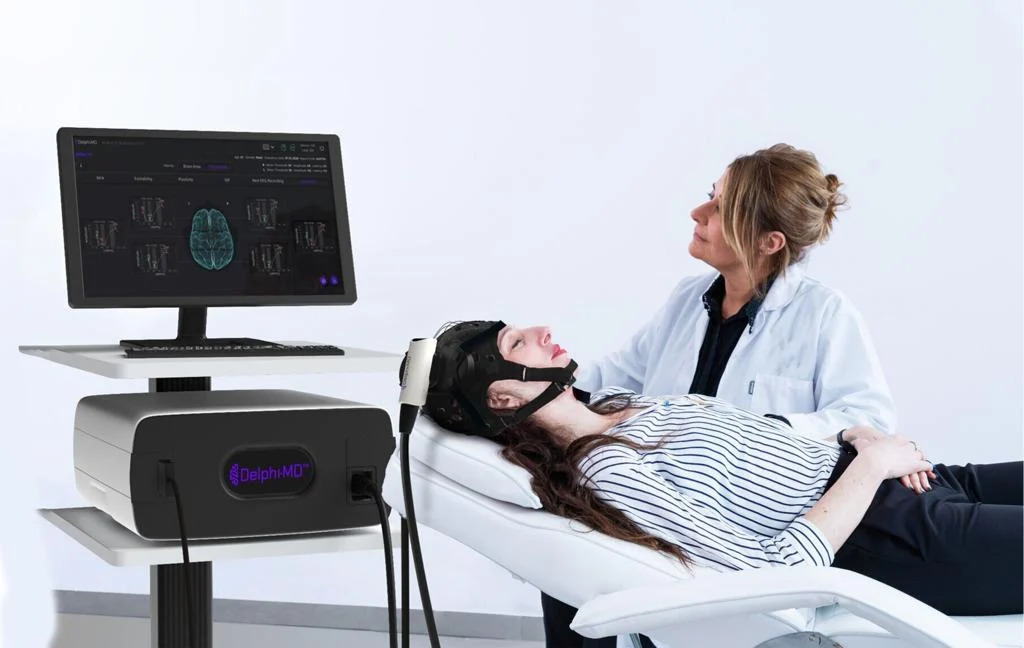
The FDA’s recent approvals aren’t limited to cardiology. QuantalX Neuroscience received De Novo classification for its Delphi-MD™ System, a novel functional neuro-imaging (FNI) technology that combines Transcranial Magnetic Stimulation (TMS) with electroencephalography (EEG) to benchmark brain network function. This provides physicians with a new tool for assessing brain health and monitoring neurological conditions.
For chronic pain sufferers, Magstim Magnetic Stimulation has been cleared by the FDA, offering a non-invasive, drug-free treatment option. This technology modulates peripheral nerve pathways, providing deeper stimulation than traditional methods.
Precision Surgery and Robotics
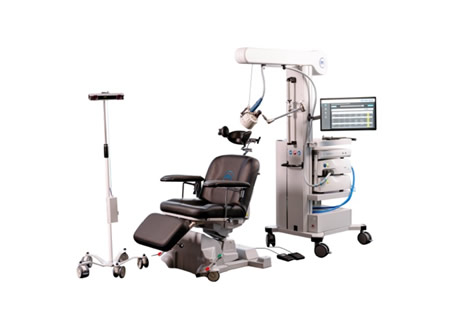
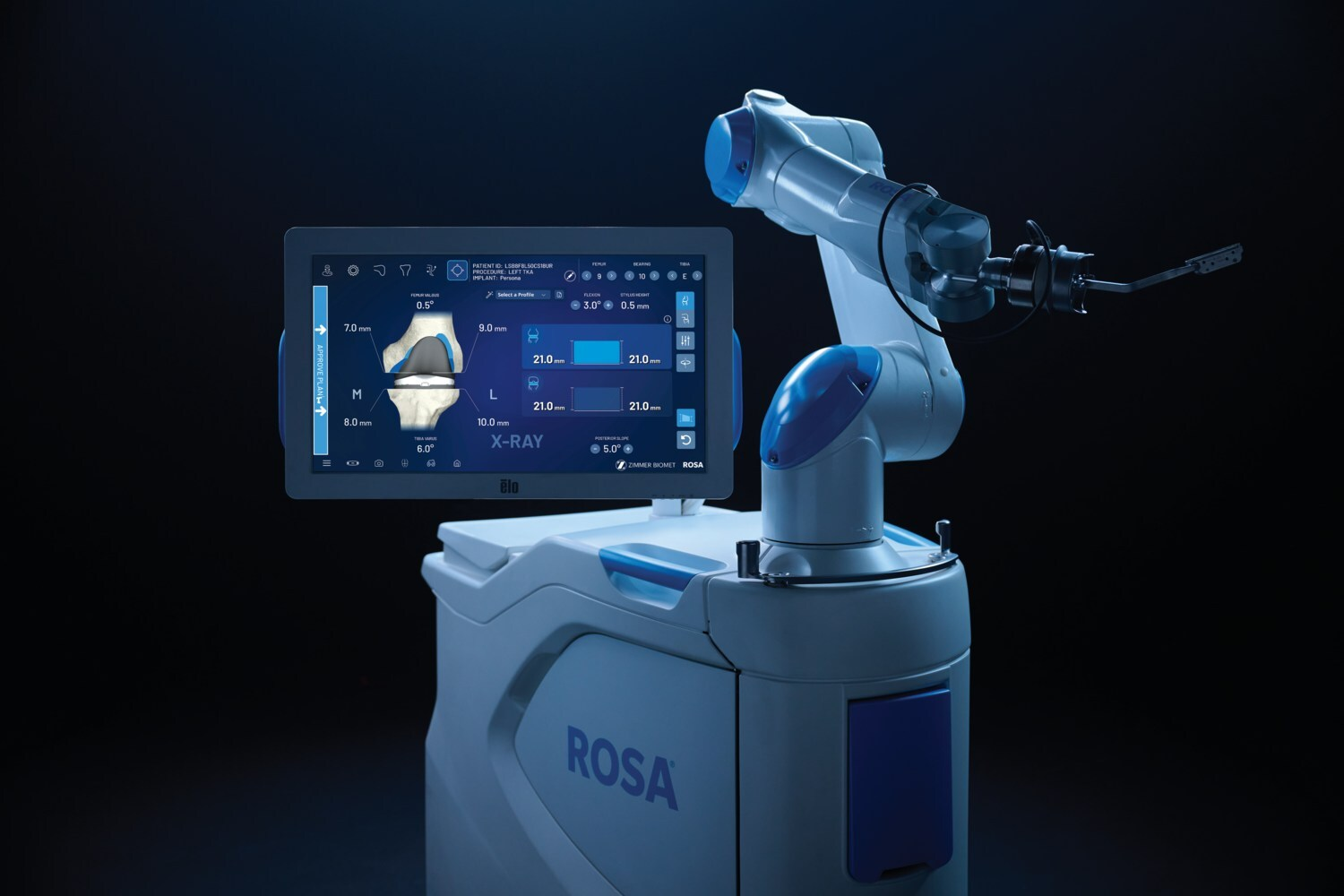
Innovations in surgical robotics are also gaining momentum. Zimmer Biomet’s ROSA® Knee with OptimiZe™, an enhanced robotic system for total knee replacement, received 510(k) clearance. This technology offers customized surgical planning and automated kinematic alignment, ensuring accurate and reproducible outcomes.
Minimally invasive surgery is also benefiting from advancements. Levita® Magnetics secured FDA clearance for its Magnetic Surgical System (MSS) for pediatric patients, utilizing external magnets to control surgical instruments and reduce the number of incisions needed. This is particularly beneficial for children, minimizing trauma and accelerating recovery.
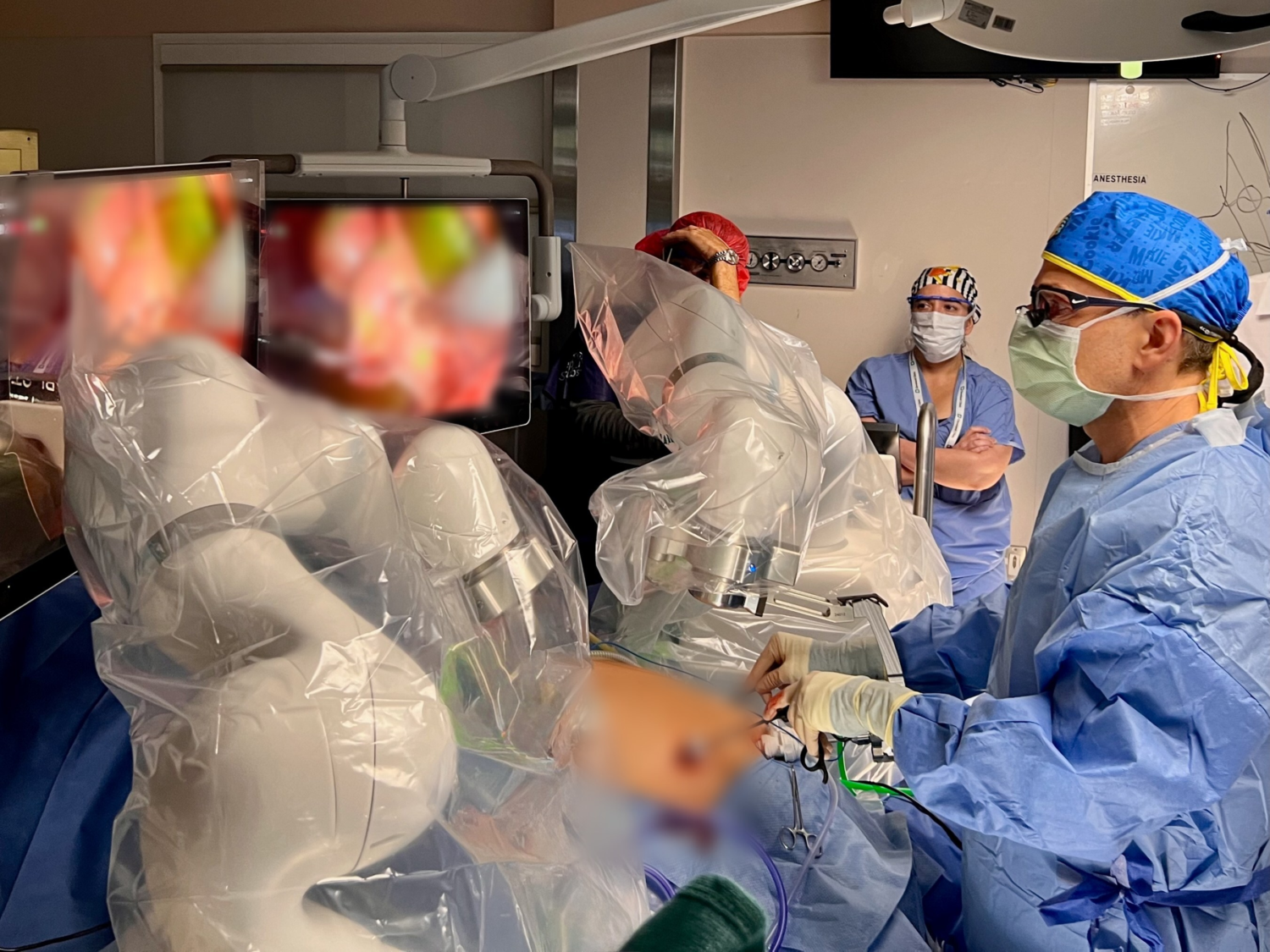
In a groundbreaking development, MMI (Medical Microinstruments, Inc.) received FDA Investigational Device Exemption (IDE) approval for a clinical study using the Symani® Surgical System for a novel microsurgical intervention for Alzheimer’s disease. This study aims to reestablish lymphatic drainage pathways in the brain, potentially improving the clearance of harmful proteins.

Addressing Chronic Conditions: Wound Care and Diabetes
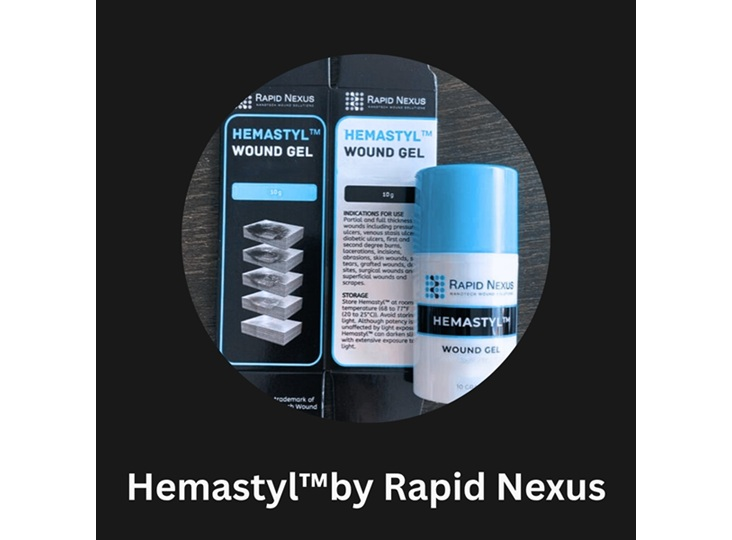
Rapid Nexus Nanotech Wound Solutions, Inc. received FDA 510(k) clearance for its Hemastyl gel device, the first to directly address the periwound tissue environment to promote wound closure and prevent amputations. This represents a paradigm shift in wound healing, moving beyond symptomatic treatment to address the underlying causes.
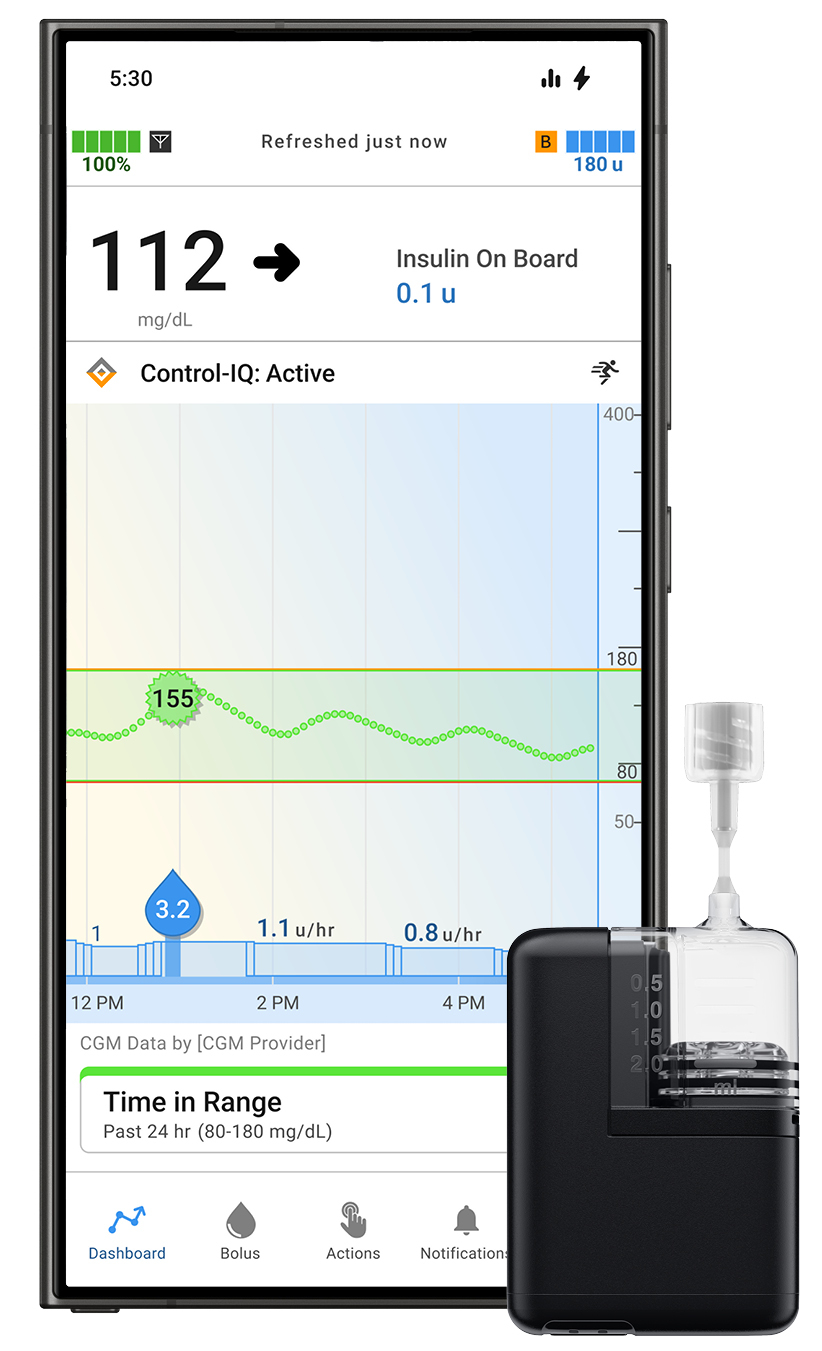
For individuals managing diabetes, Tandem Diabetes Care, Inc. has received FDA clearance for the Android version of its Tandem Mobi app, expanding access to its automated insulin delivery system powered by Control-IQ+ technology.
What impact will these advancements have on the future of personalized medicine? And how will healthcare systems adapt to integrate these new technologies into existing workflows?
Frequently Asked Questions About Recent FDA Clearances
What is the significance of these recent FDA clearances?
These clearances represent a major leap forward in medical technology, driven by the increasing integration of artificial intelligence and advanced diagnostics. They promise to improve patient outcomes, reduce invasiveness, and expand access to care.
How will AI-powered diagnostics impact the role of physicians?
AI is not intended to replace physicians, but rather to augment their capabilities. These tools can assist with complex analyses, reduce cognitive burden, and provide more accurate insights, allowing doctors to focus on patient interaction and personalized treatment plans.
What are the benefits of wearable medical devices like Hexoskin?
Wearable devices enable continuous, long-term monitoring of vital signs outside of traditional clinical settings. This provides a more comprehensive understanding of a patient’s health and allows for earlier detection of potential problems.
How does the RapidAI Aortic Management system improve patient care?
RapidAI’s system automates critical measurements and provides 3D reconstructions of the aorta, aiding in the diagnosis and management of aortic disease. This leads to faster read times, improved accuracy, and more informed treatment decisions.
What is the potential of the Delphi-MD system for neurological conditions?
The Delphi-MD system offers a novel approach to functional neuro-imaging, providing a benchmarked assessment of brain function. This can aid in the monitoring of cognitive decline, evaluation of neurological interventions, and management of various neurological conditions.
How does the Levita Magnetic Surgical System benefit pediatric patients?
The MSS utilizes external magnets to control surgical instruments, reducing the number of incisions needed for procedures. This minimizes trauma, scarring, and recovery time, which is particularly important for children.
Disclaimer: This article provides general information about recent FDA clearances and should not be considered medical advice. Consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.

