Glioblastoma, the most aggressive form of brain cancer, has long defied effective treatment. While immunotherapy has revolutionized cancer care for many tumor types, glioblastoma has remained stubbornly resistant. Now, a groundbreaking study from MIT’s Koch Institute offers a crucial new understanding of *why* this resistance exists – and, more importantly, identifies potential pathways to overcome it. This isn’t just another incremental step; it’s a fundamental shift in how we approach this devastating disease, moving beyond simply trying to activate the immune system to understanding the complex interplay between cancer cells and the immune environment they create.
- The Macrophage Problem: Glioblastoma hijacks macrophages – immune cells meant to fight cancer – and turns them into protectors of the tumor, suppressing T cell activity.
- Co-Evolution is Key: Researchers discovered that both glioblastoma cells and macrophages change in predictable ways when they interact, revealing new therapeutic targets.
- mRNA Therapy Shows Promise: Targeting specific antigens identified through this research led to significant tumor reduction and even eradication in mouse models.
For decades, glioblastoma treatment has followed a standard protocol: surgery, followed by radiation and chemotherapy. Despite this aggressive approach, the prognosis remains grim – less than half of patients survive beyond 15 months, and only 5% live longer than five years. The failure of immune checkpoint inhibitors, a class of drugs that have shown remarkable success in other cancers, underscored the unique challenges posed by glioblastoma. The problem isn’t that the patient’s immune system is inherently unable to fight the cancer; it’s that the tumor actively suppresses the immune response, specifically by recruiting and reprogramming macrophages.
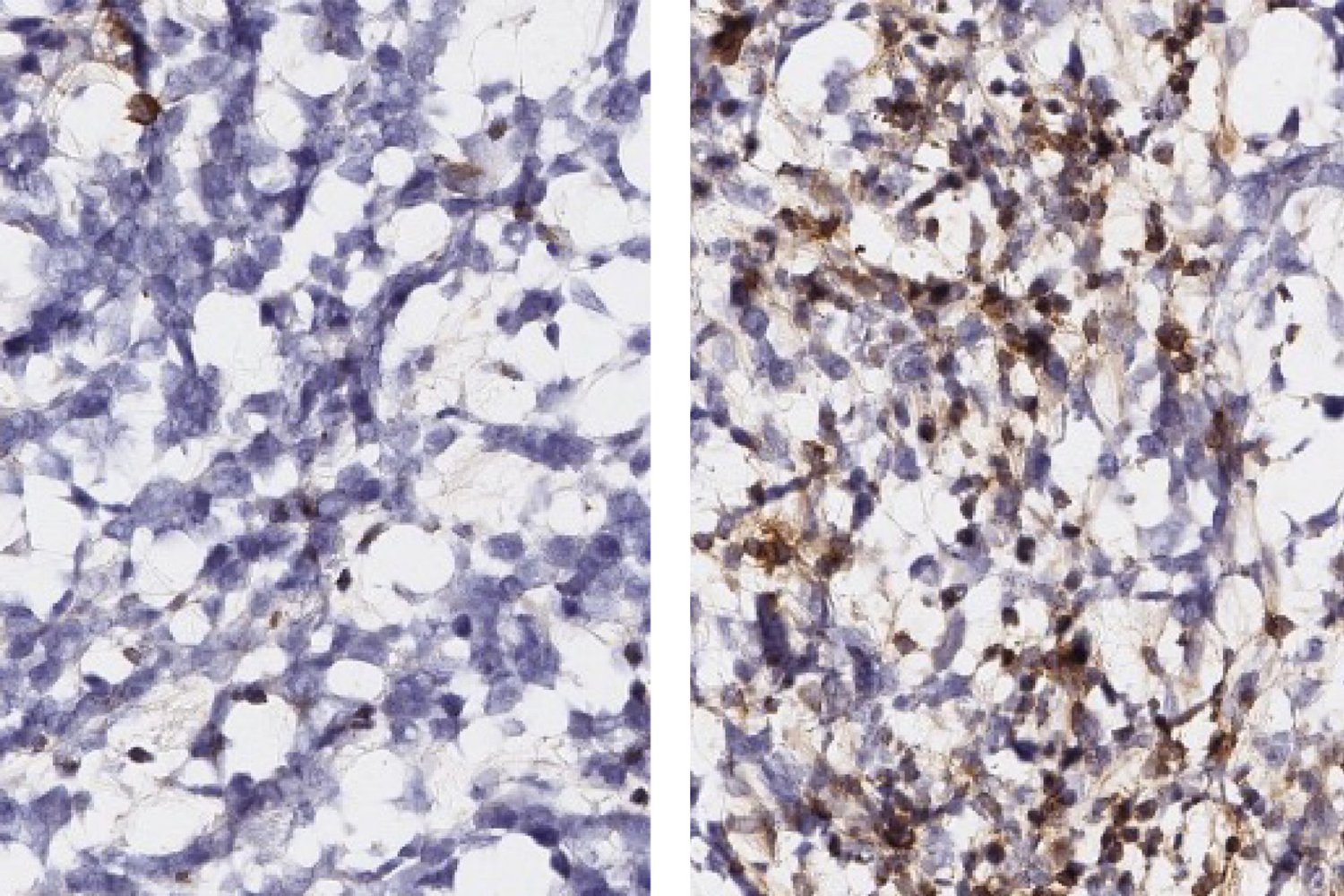
The MIT team, led by Forest White, didn’t just look at the cancer cells or the macrophages in isolation. They employed sophisticated “immune profiling tools” to map out the dynamic relationship between the two – a “co-evolution,” as White describes it. This approach, drawing an analogy to how a new family changes a neighborhood, revealed that the interaction fundamentally alters both cell types. By analyzing the “immunopeptidome” – the antigens presented on the surface of these cells – researchers identified over 800 peptides in macrophages that changed expression when co-cultured with glioblastoma cells. Critically, these changes pointed to cytokine signaling pathways that promote tumor aggression and suppress the immune response.
The study also revealed changes in antigen presentation on the glioblastoma cells themselves, linked to the Rho GTPase signaling protein, a known driver of cancer development. Importantly, the patterns observed in laboratory cell cultures were validated in mouse models and, potentially, in human tumor samples, strengthening the relevance of these findings.
The Forward Look
The identification of six key antigens as therapeutic targets is the most immediate and exciting outcome of this research. The success of mRNA-based immunostimulatory therapies in mouse models suggests a viable path forward for clinical translation. However, several hurdles remain. The next critical step will be to validate these targets in larger animal models and, ultimately, in human clinical trials. Furthermore, the team’s plan to extend their immunopeptidome profiling to dendritic cells – the key antigen-presenting cells that bridge the gap between cancer and T cell activation – is crucial. Understanding how glioblastoma influences dendritic cell function could unlock even more potent immunotherapeutic strategies.
Beyond glioblastoma, the methodology developed by the White lab – quantitative, cell-type-specific immunopeptidome profiling – has broad implications for cancer research. As Cui notes, this approach could be applied to design improved immunotherapies for a wide range of cancers and other diseases. We can expect to see increased investment in this type of sophisticated immune profiling as researchers strive to overcome the challenges of tumor immune evasion. This study isn’t just about a new treatment for glioblastoma; it’s about a new way of *thinking* about cancer immunotherapy, one that prioritizes understanding the complex interactions within the tumor microenvironment.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.