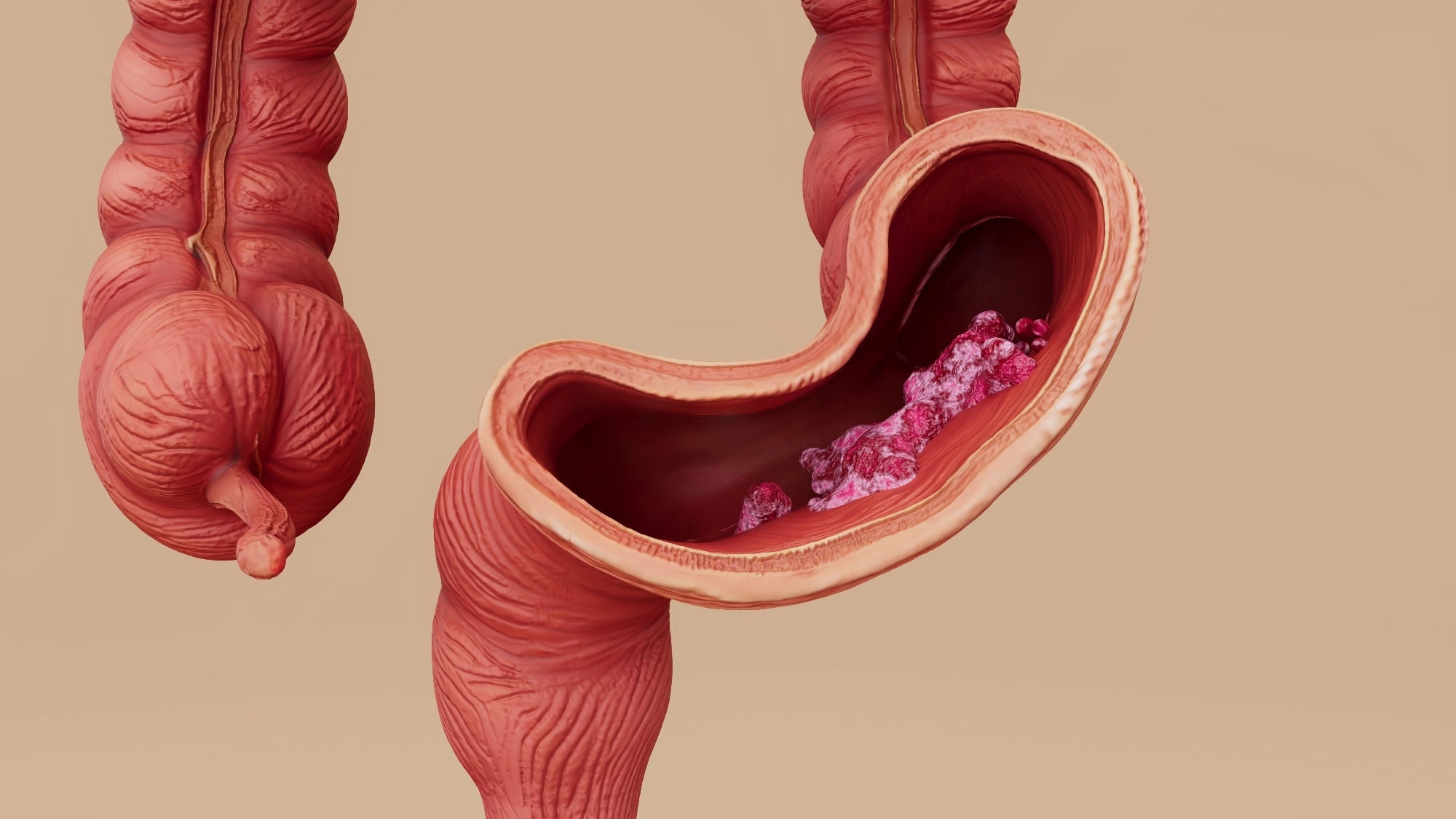
Rising rates of gastrointestinal cancers, even in younger populations, are no longer a statistical anomaly – they’re a signal of a deeper, systemic shift in how we understand cancer development. This isn’t simply about genetics or lifestyle; it’s about the intricate, and often overlooked, relationship between the trillions of microorganisms residing in our gut and our overall health. New research is solidifying the gut microbiome’s role not just as a bystander, but as an active participant in tumor initiation, progression, and even response to therapy. This understanding is poised to revolutionize preventative strategies and treatment paradigms for colorectal and gastric cancers.
- The Microbiome as a Driver: Gut dysbiosis – an imbalance in microbial communities – fuels chronic inflammation and directly damages DNA, increasing cancer risk.
- H. pylori’s Long Shadow: Helicobacter pylori remains a critical factor in gastric cancer, but the interplay with other gut microbes is now recognized as equally important.
- Therapeutic Potential: Targeting the microbiome through interventions like FMT and tailored diets is emerging as a promising adjunct to conventional cancer treatments.
For decades, cancer research focused primarily on the genetic mutations within tumor cells. However, the emerging field of microbiome research reveals a far more complex picture. The gut microbiome, a dynamic ecosystem of bacteria, viruses, fungi, and other microorganisms, isn’t merely a passive inhabitant of our digestive system. It actively communicates with our cells, influencing immune responses, metabolic processes, and even gene expression. The recent surge in early-onset GI cancers, particularly among racial and ethnic minorities, underscores the need to investigate environmental factors – and the microbiome is a prime candidate.
Dysbiosis, characterized by a loss of microbial diversity and an overgrowth of harmful bacteria, creates a pro-inflammatory environment. This chronic inflammation damages the intestinal lining, disrupts immune function, and provides fertile ground for tumor development. Specific bacterial strains, like Fusobacterium nucleatum and enterotoxigenic Bacteroides fragilis, produce toxins that directly damage DNA, while others manipulate signaling pathways to promote cell growth and suppress immune responses. The discovery of microbial metabolites – the byproducts of bacterial metabolism – further complicates the picture. While some metabolites, like short-chain fatty acids (SCFAs), can be protective, others contribute to inflammation and DNA damage.
The case of Helicobacter pylori and gastric cancer serves as a stark example of the microbiome’s carcinogenic potential. While H. pylori infection has long been linked to gastric cancer, research now shows that the subsequent alterations to the gastric microbiome – the “non-H. pylori acceleration” cascade – are equally crucial. This highlights the importance of considering the entire microbial ecosystem, not just a single pathogen.
The Forward Look
The implications of these findings are far-reaching. We are on the cusp of a new era of personalized cancer prevention and treatment, where the microbiome is a central target. The development of sophisticated diagnostic tools – leveraging metagenomics and metabolomics – will allow for early detection of dysbiosis and identification of specific microbial signatures associated with increased cancer risk. This will pave the way for targeted interventions, such as personalized dietary recommendations, prebiotic/probiotic therapies, and even fecal microbiota transplantation (FMT).
However, significant challenges remain. The microbiome is incredibly complex and varies significantly between individuals. Standardizing analytical methods and conducting large-scale, longitudinal studies are crucial to overcome this variability and translate research findings into clinical practice. Furthermore, the interplay between the microbiome, genetics, and lifestyle factors is still poorly understood. Expect to see increased investment in multi-omics research – integrating genomics, metabolomics, and microbiome data – to unravel these complex interactions. The next few years will likely see clinical trials evaluating the efficacy of microbiome-targeted therapies in combination with conventional cancer treatments, potentially offering a new avenue for improving patient outcomes and reducing the burden of gastrointestinal cancers.
Last Updated: Feb 19, 2026
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.