Lab-Grown Organs: The Dawn of Personalized Transplants and a $300 Billion Market
Over 100,000 people in the United States alone are currently waiting for organ transplants. Every ten minutes, another name is added to the list. But what if the agonizing wait, the constant risk of rejection, and the ethical dilemmas surrounding donor organs could be drastically reduced – or even eliminated? Recent breakthroughs in bioengineering, specifically the successful implantation of a lab-grown esophagus in animal models, suggest we are on the cusp of a revolution in regenerative medicine. This isn’t just about solving the organ shortage; it’s about ushering in an era of personalized transplants, tailored to each patient’s unique biological needs.
The Esophagus Breakthrough: A Milestone, Not an Isolated Event
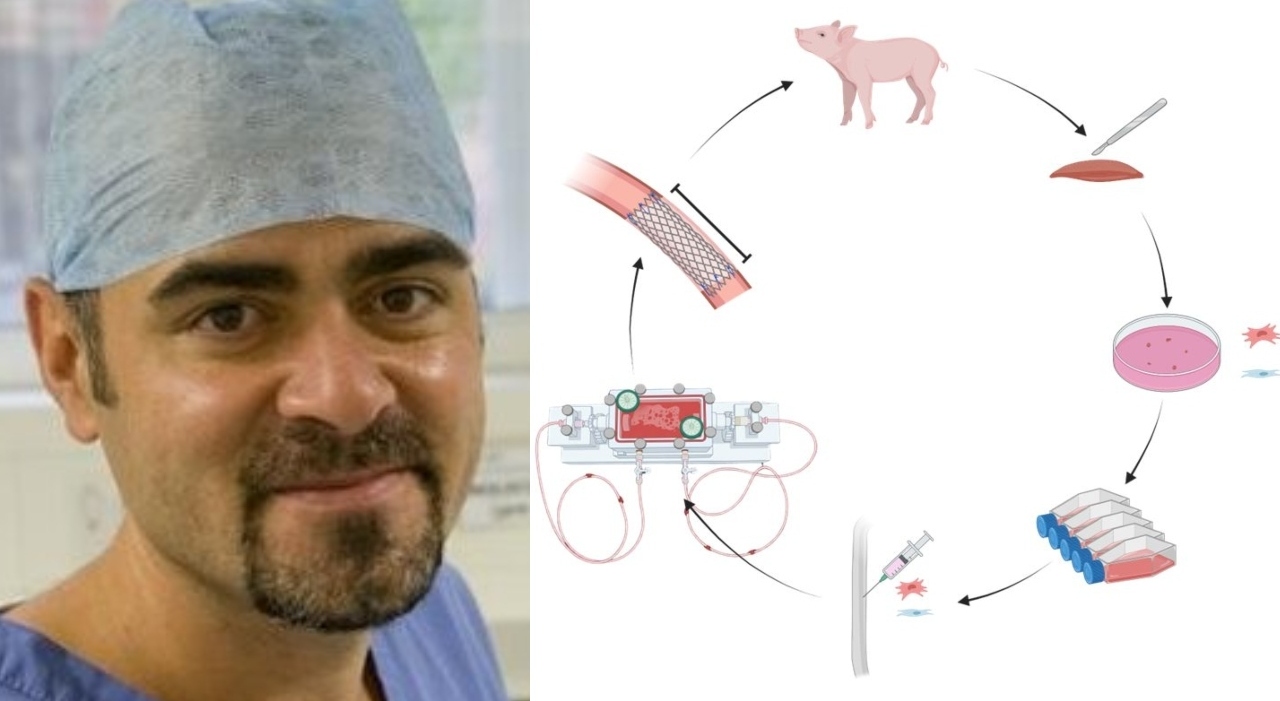
Italian researchers, as reported by Leggo.it, Corriere della Sera, and ANSA, have achieved a significant milestone: a functional esophagus created from stem cells and successfully implanted into pigs. While animal trials are a crucial first step, the implications are profound. This success builds upon years of research into bioprinting, decellularization, and stem cell differentiation – techniques that are rapidly maturing and expanding beyond the esophagus.
Beyond the Esophagus: A Pipeline of Lab-Grown Organs
The esophagus is just the beginning. Scientists are actively working on growing a wide range of organs in the lab, including kidneys, livers, hearts, and even lungs. Decellularization, the process of stripping an organ of its cells leaving behind a collagen scaffold, is proving particularly effective. This scaffold can then be repopulated with a patient’s own cells, minimizing the risk of rejection. Bioprinting, using 3D printing techniques to layer cells and biomaterials, offers even greater precision and control over organ structure.
The Economic and Ethical Landscape of Bioengineered Organs
The potential market for lab-grown organs is staggering. Analysts predict a $300 billion market by 2040, driven by the increasing demand for transplants and the promise of personalized medicine. However, this growth isn’t without its challenges. The cost of production remains high, and scaling up manufacturing to meet global demand will require significant investment and innovation. Ethical considerations, such as equitable access to these life-saving technologies and the potential for misuse, must also be addressed proactively.
Addressing the Cost Barrier: From Luxury to Accessibility
Currently, the cost of creating a single lab-grown organ is prohibitive, potentially exceeding hundreds of thousands of dollars. However, advancements in bioreactor technology, automation, and cell sourcing are driving down costs. Furthermore, the long-term economic benefits – reduced healthcare costs associated with chronic organ failure and immunosuppressant drugs – could offset the initial investment. Government funding, public-private partnerships, and innovative financing models will be crucial to making these therapies accessible to all who need them.
The Future of Transplantation: A Personalized, On-Demand System
Imagine a future where organ rejection is a relic of the past. Where patients receive organs grown from their own cells, eliminating the need for lifelong immunosuppression. Where organ shortages are a distant memory, and the waiting list is a thing of the past. This isn’t science fiction; it’s a rapidly approaching reality. The convergence of stem cell research, bioprinting, and advanced biomaterials is poised to transform transplantation as we know it.
| Organ | Current Status (Feb 2024) | Projected Timeline for Clinical Trials |
|---|---|---|
| Esophagus | Successful animal trials | Human trials within 3-5 years |
| Kidney | Partial kidney structures grown; decellularization techniques advanced | Early-stage human trials within 5-7 years |
| Liver | Functional liver tissue engineered; challenges remain in vascularization | Preclinical studies ongoing; 7-10 years to clinical trials |
| Heart | Significant progress in cardiac patch development; full organ creation complex | Long-term research; 10+ years to clinical trials |
Frequently Asked Questions About Lab-Grown Organs
What are the biggest hurdles to widespread adoption of lab-grown organs?
The primary challenges include scaling up production to meet demand, reducing costs, ensuring long-term organ functionality, and navigating complex regulatory pathways.
Will lab-grown organs be available to everyone, or will they be limited to the wealthy?
Ensuring equitable access is a critical concern. Government funding, public-private partnerships, and innovative financing models will be essential to make these therapies affordable and accessible to all.
How does the ethical debate surrounding stem cell research impact the development of lab-grown organs?
Ethical considerations surrounding stem cell sourcing and potential misuse are ongoing. Strict regulations and ethical guidelines are necessary to ensure responsible development and application of these technologies.
What role will 3D bioprinting play in the future of organ creation?
3D bioprinting offers unprecedented precision and control over organ structure, allowing for the creation of complex tissues and organs tailored to individual patients. It’s a key technology driving the field forward.
The future of transplantation is not about waiting for a donor; it’s about creating the organs we need, on demand. What are your predictions for the impact of lab-grown organs on healthcare in the next decade? Share your insights in the comments below!
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.