Libtayo Approved in EU: A New Adjuvant Immunotherapy Option for Cutaneous Squamous Cell Carcinoma
Brussels, Belgium – In a landmark decision for patients facing high-risk cutaneous squamous cell carcinoma (CSCC), the European Commission has granted marketing authorization for Libtayo® (cemiplimab). This approval marks a significant advancement, establishing Libtayo as the first and only immunotherapy specifically indicated for adjuvant treatment following surgical resection and/or radiation therapy in individuals with CSCC at substantial risk of recurrence. This breakthrough offers a new hope for improved outcomes and a potentially better quality of life for those affected by this aggressive skin cancer.
Understanding Cutaneous Squamous Cell Carcinoma and the Role of Immunotherapy
Cutaneous squamous cell carcinoma (CSCC) is a common form of skin cancer, arising from the squamous cells that make up the outer layer of the skin. While often treatable with surgery or radiation, a significant proportion of patients experience recurrence, particularly those with high-risk features such as aggressive tumor biology, perineural invasion, or involvement of specific anatomical sites. Traditionally, managing this risk involved close surveillance and further aggressive local treatments upon recurrence.
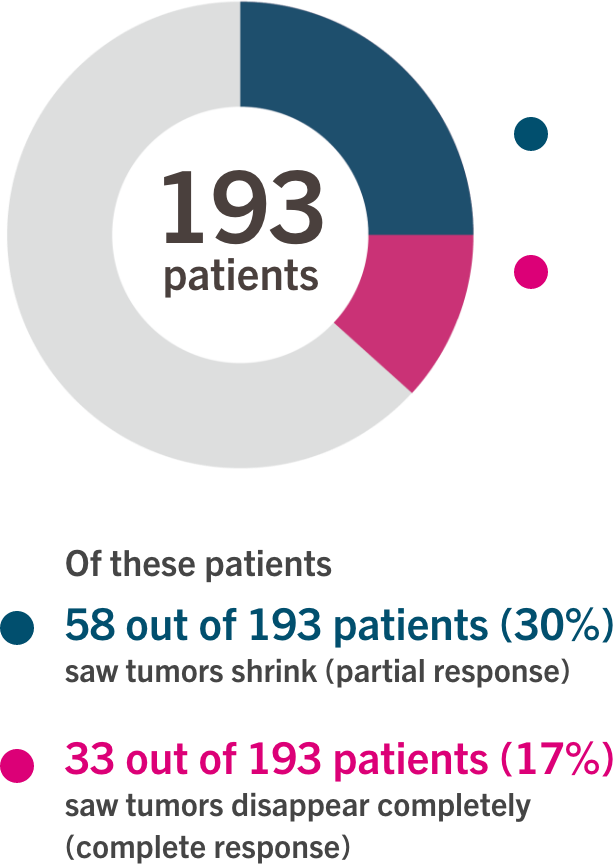
Immunotherapy, a revolutionary approach to cancer treatment, harnesses the power of the body’s own immune system to fight cancer cells. Libtayo, a human monoclonal antibody targeting the PD-1 receptor, works by blocking the interaction between PD-1 and its ligands, PD-L1 and PD-L2. This blockade releases the brakes on the immune system, allowing T cells to recognize and attack cancer cells more effectively. The approval is based on data from the pivotal Phase 3 LIBERTY-248 trial, which demonstrated a statistically significant and clinically meaningful reduction in the risk of recurrence or death compared to placebo.
A critical aspect of post-surgical CSCC management is understanding the potential for second primary tumors. Pharmacy Times provides a detailed exploration of these tumors, their definitions, incidence, and clinical implications in the context of cemiplimab treatment. Recognizing and addressing these secondary cancers is crucial for comprehensive patient care.
The European Commission’s decision follows a similar approval in the United States, further solidifying Libtayo’s position as a leading immunotherapy option for CSCC. Regeneron, the manufacturer of Libtayo, highlighted the potential of this therapy to transform the treatment landscape for patients with high-risk CSCC. Investing.com also reported on the approval, emphasizing its significance for European patients.
What impact do you foresee this approval having on the standard of care for CSCC patients across Europe? And how might this advancement influence the development of future immunotherapies for other types of skin cancer?
Frequently Asked Questions About Libtayo and CSCC
-
What is Libtayo and how does it treat cutaneous squamous cell carcinoma?
Libtayo (cemiplimab) is an immunotherapy that blocks the PD-1 receptor, enabling the immune system to attack cancer cells. It’s approved as an adjuvant treatment for high-risk CSCC after surgery or radiation.
-
Who is eligible for Libtayo treatment for CSCC?
Libtayo is indicated for patients with cutaneous squamous cell carcinoma who have undergone surgical resection and/or radiation therapy and are at high risk of recurrence.
-
What are the potential side effects of Libtayo?
Like all immunotherapies, Libtayo can cause immune-related adverse events. These can affect various organs and require careful monitoring and management by a healthcare professional.
-
How does adjuvant immunotherapy differ from other CSCC treatments?
Adjuvant immunotherapy, like Libtayo, is given *after* primary treatment (surgery or radiation) to reduce the risk of the cancer returning. It differs from treatments aimed at directly shrinking the tumor.
-
What is the significance of the LIBERTY-248 trial in the approval of Libtayo?
The LIBERTY-248 trial provided the clinical evidence demonstrating that Libtayo significantly reduces the risk of recurrence or death in high-risk CSCC patients, leading to its approval.
Disclaimer: This article provides general information and should not be considered medical advice. Always consult with a qualified healthcare professional for diagnosis and treatment of any medical condition.
Share this important news with your network and join the conversation below. What are your thoughts on the future of immunotherapy in cancer treatment?
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.