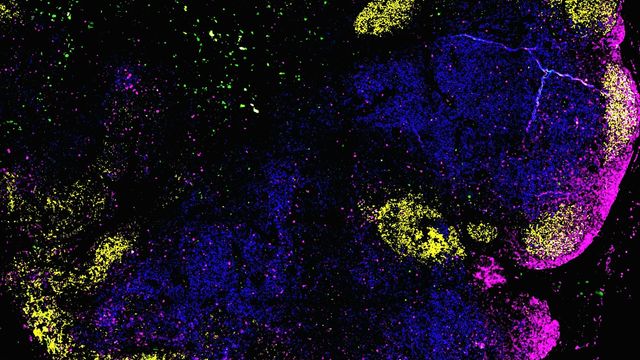
The intricate architecture of our lymphatic system, long understood to be vital for immune function, is now revealed as a critical battleground in the fight against lymphoma. Groundbreaking research from a collaborative team led by Heinrich Heine University Düsseldorf (HHU) has, for the first time, mapped the organization of immune cells within human lymph nodes and pinpointed how this structure degrades in malignant lymphomas – a discovery with significant implications for prognosis and treatment strategies. This isn’t simply about understanding *how* lymphoma progresses; it’s about understanding *why* certain patients fare worse than others, and how we can intervene to alter that trajectory.
- Lymph Node Architecture Matters: The study confirms that the spatial organization of immune cells within lymph nodes directly impacts survival rates in lymphoma patients.
- Inflammation is a Key Driver: A previously underestimated inflammatory cycle actively contributes to the breakdown of lymph node structure, rather than being a passive consequence of tumor growth.
- New Therapeutic Avenues: Stabilizing stromal cells or modulating inflammatory signals within the tumor microenvironment presents a promising new approach to lymphoma treatment.
For decades, researchers have known that lymph nodes – the hubs of our immune defenses – rely on a precise arrangement of B cells, T cells, and supporting stromal cells. These stromal cells act as “architects,” releasing chemokines that guide immune cells to their designated locations. The study highlights that this organization isn’t static. In aggressive lymphomas like diffuse large B-cell lymphoma (DLBCL), this carefully constructed architecture collapses, while slower-growing lymphomas, such as follicular lymphoma (FL), exhibit a more subtle disruption. Until now, the underlying mechanisms driving these differing patterns of degradation remained elusive. The increasing incidence of lymphoma globally, coupled with the limitations of current treatment options, has made understanding these mechanisms a critical priority.
The HHU-led team employed cutting-edge single-cell analysis and spatial tissue mapping to reveal that changes in chemokine signaling within stromal cells are central to this process. Specifically, they identified an inflammatory vicious cycle: tumor-associated T cells release interferons, which reprogram stromal cells to produce inflammatory chemokines instead of structural signals. This attracts more inflammatory cells, further disrupting the lymph node’s organization. This is a crucial finding because it reframes the understanding of lymph node disruption – it’s not merely a result of the cancer’s presence, but an actively driven process.
The Forward Look
The implications of this research extend far beyond a deeper understanding of lymphoma biology. The identification of specific chemokine signatures associated with poor prognosis opens the door to earlier, more accurate disease stratification. We can anticipate a surge in research focused on developing biomarkers based on these findings, potentially leading to companion diagnostics that predict treatment response. More importantly, the study validates the tumor microenvironment – and specifically, stromal cells – as a viable therapeutic target. Expect to see increased investment in therapies designed to stabilize stromal function or modulate inflammatory signaling pathways within lymph nodes. Clinical trials exploring these approaches are likely to emerge within the next 3-5 years. Furthermore, the techniques used in this study – single-cell analysis and spatial tissue mapping – are becoming increasingly accessible, and will undoubtedly be applied to other cancers to unravel the complex interplay between tumor cells and their microenvironment. The era of precision oncology is increasingly focused on the ecosystem *around* the tumor, and this research is a significant step in that direction.
Reference: Czernilofsky F, Mathioudaki A, Jopp-Saile L, et al. Reprogramming of stroma-derived chemokine networks drives the loss of tissue organization in nodal B cell lymphoma. Nat Cancer. 2026. doi: 10.1038/s43018-026-01136-z
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.