The secret to managing cellular metabolism may not lie in complex human trials, but in the growth cycle of a common weed. New research into the protein PEX11 has revealed a universal mechanism for controlling peroxisome size—the cellular “power plants” responsible for breaking down fatty acids—marking a significant step forward in our ability to manipulate cell architecture for both medical and industrial purposes.
- The Regulator: PEX11 protein was identified as the primary controller for the expansion and shrinkage of peroxisomes during critical developmental windows.
- CRISPR Precision: Researchers bypassed genetic redundancy (five different PEX11 genes) using advanced CRISPR techniques to isolate the protein’s function without killing the organism.
- Cross-Species Utility: The discovery that yeast PEX11 can restore function in plant cells suggests this mechanism is “highly conserved,” meaning it likely operates similarly in human cells.
The Deep Dive: Why This Matters
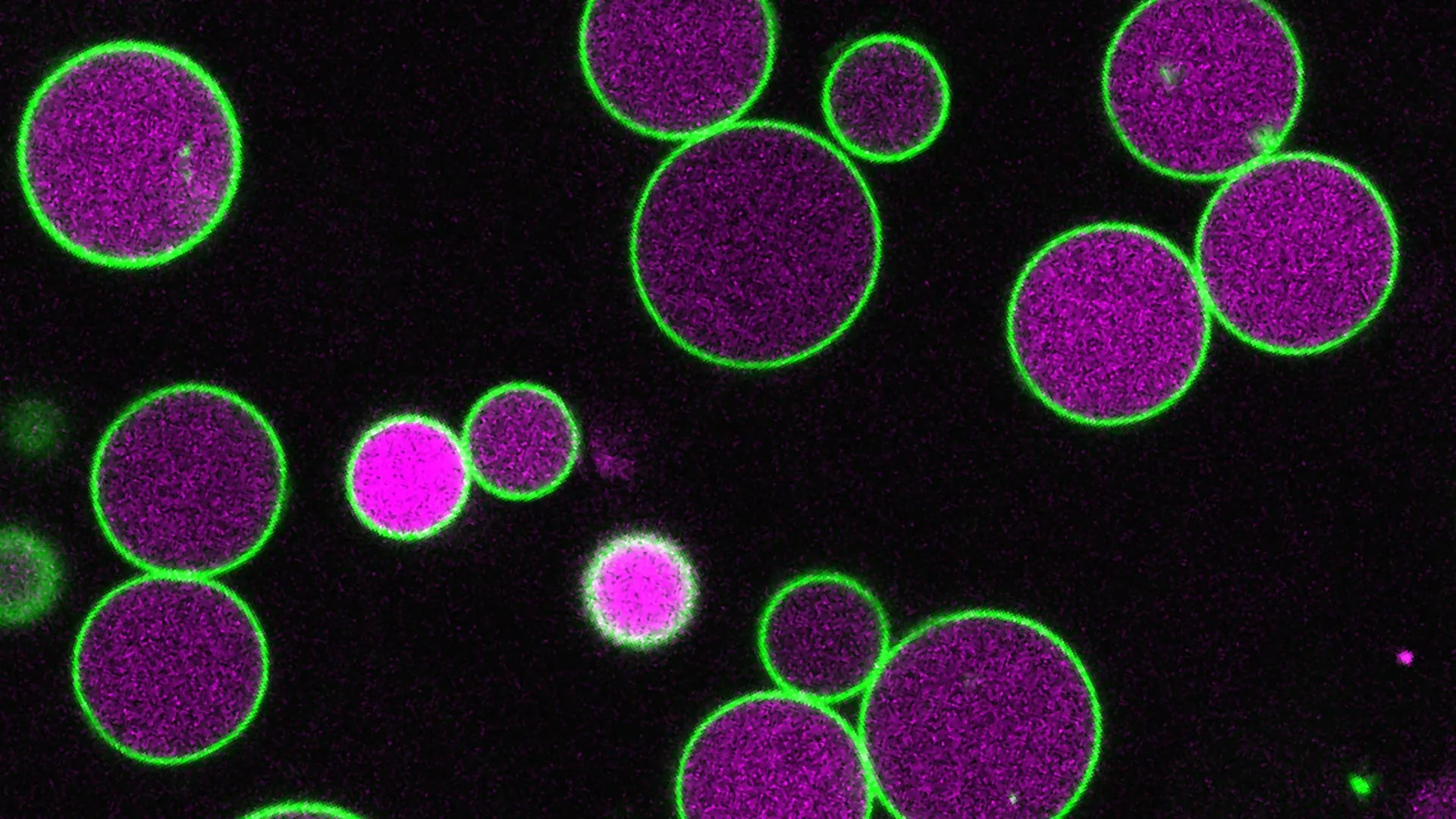
To understand the significance of this find, one must understand the peroxisome. These membrane-bound compartments are essential for metabolic flexibility. In plants, they are the bridge between a seed’s stored energy (fatty acids) and its adult life (photosynthesis). If a plant cannot transition its peroxisome size effectively, it fails to thrive. For researchers, the Arabidopsis plant serves as a living laboratory because its peroxisomes are large enough to be observed under a standard light microscope, providing a visual clarity that human cells simply cannot offer.
The technical hurdle in this research was genetic redundancy. Nature rarely relies on a single point of failure; in this case, five different genes produced PEX11. Traditional “knockout” methods—disabling one gene—did nothing, while disabling all five was lethal. By using CRISPR to selectively break specific combinations, Nathan Tharp and his team were able to create “giant” peroxisomes that stretched across entire cells. This revealed the hidden mechanism: PEX11 facilitates the creation of internal vesicles that “prune” the outer membrane. Without PEX11, the peroxisome grows unchecked, like a balloon that cannot leak air.
The Forward Look: Beyond the Greenhouse
While the study used plants, the “tech” here is the conservation of the protein. The fact that yeast PEX11—separated by millions of years of evolution—works in a plant cell is a massive signal for bioengineers. We are no longer looking at a “plant quirk,” but a fundamental rule of cellular biology.
From a strategic standpoint, the next steps are clear: Precision Bioengineering. If we can control the size and efficiency of peroxisomes, we can potentially optimize cells used in industrial fermentation to produce biofuels or pharmaceuticals more efficiently. More critically, because peroxisomes are implicated in various human metabolic diseases, this research provides a roadmap for targeting PEX11-like proteins to treat cellular dysfunction in humans.
Watch for upcoming studies that attempt to apply these CRISPR-driven insights to mammalian cells. The jump from Arabidopsis to humans is steep, but the underlying “code” for peroxisome regulation appears to be the same.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.