The materials science world just took a significant, if quiet, leap forward. Researchers in China have created the first polymer networks utilizing ‘pnictogen bonds’ – a relatively newly defined type of chemical interaction – to create materials with controllable self-healing properties, even underwater. This isn’t just a lab curiosity; it addresses a critical limitation in current self-healing materials and opens doors to more robust and adaptable technologies.
- Beyond Hydrogen: This research moves beyond traditional hydrogen bonding, which is often too weak and solvent-sensitive for demanding applications.
- Underwater Healing: The key breakthrough is maintaining self-healing capability *in aqueous environments* – a major hurdle for many existing self-healing polymers.
- Tunable Properties: By adjusting the antimony’s oxidation state, researchers can fine-tune the strength of the bonds and, consequently, the material’s properties.
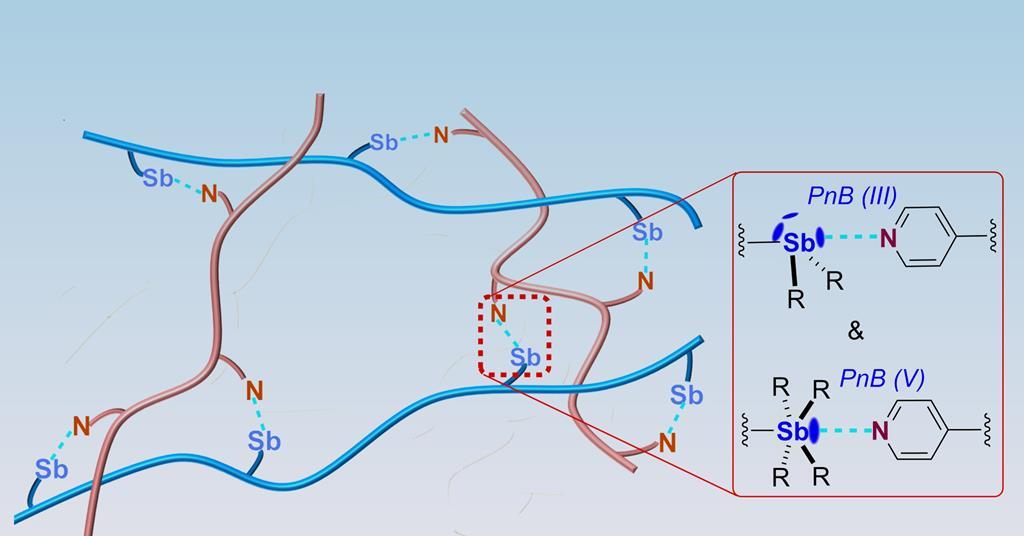
For years, materials scientists have been chasing the holy grail of truly self-healing materials. The promise is immense: longer-lasting products, reduced waste, and increased safety in critical applications. Traditional approaches rely on covalent bonds (strong but inflexible) or supramolecular interactions like hydrogen bonds (weak and easily disrupted). While halogen and chalcogen bonding have shown promise, they haven’t offered the ideal balance of strength, reversibility, and environmental tolerance. The recent formal definition of pnictogen bonding – interactions involving elements like nitrogen, phosphorus, and antimony – provided a new avenue for exploration. The challenge was always translating that theoretical understanding into a functional material.
The Zhengzhou University team overcame this by leveraging the unique properties of antimony and pyridine-functionalized polymer chains. Antimony’s strong Lewis acidity creates a robust, directional bond. Crucially, the researchers demonstrated the ability to control the bond strength by altering antimony’s oxidation state (between Sb(III) and Sb(V)). This level of control is what separates this research from previous attempts at dynamic polymer materials. The accessible ‘sigma-holes’ in pnictogen bond donors allow for stronger and more predictable interactions, leading to greater control over the final material’s characteristics. And, importantly, these bonds are remarkably stable in water, a common failure point for other self-healing polymers.
The Forward Look: Don’t expect self-healing phone screens tomorrow, but this is a foundational step. The next phase will focus on scaling production and exploring different pnictogen elements beyond antimony to optimize performance and cost. We’ll likely see this technology first integrated into high-value applications where durability and reliability are paramount – think advanced coatings for infrastructure, self-sealing materials for aerospace, or even biocompatible polymers for medical implants. The biggest question mark is long-term stability. While the initial results are promising, rigorous testing over extended periods will be needed to assess the material’s lifespan and resistance to degradation. Competitors will undoubtedly begin exploring pnictogen bonding, potentially leading to a rapid acceleration of innovation in this space. Keep an eye on patent filings from materials science groups over the next 12-18 months – they’ll be a strong indicator of who is seriously pursuing this technology.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.