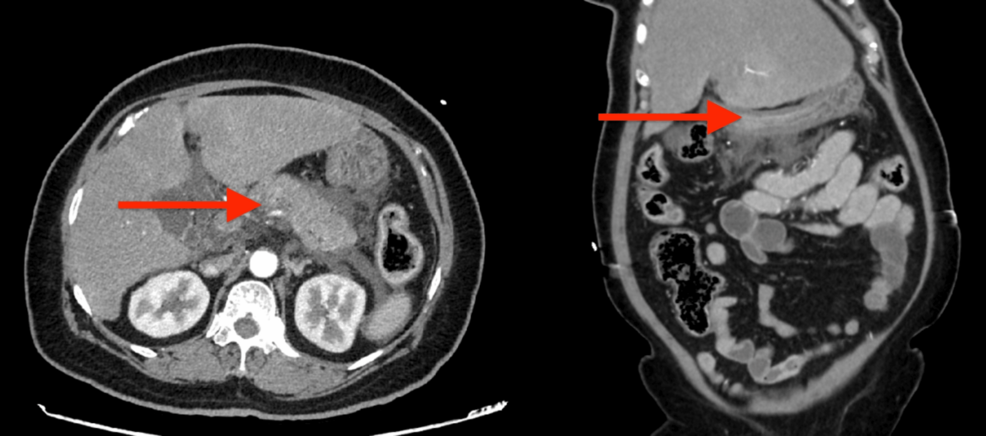
The GLP-1 Revolution: Navigating the Rising Risk of Pancreatitis and the Future of Weight Loss Drugs
Over 40 million prescriptions for GLP-1 receptor agonists – drugs like semaglutide and liraglutide – were filled in the US last year, a figure that represents a seismic shift in the treatment of both type 2 diabetes and obesity. But this rapid ascent isn’t without potential consequences. Recent case reports, including instances of acute pancreatitis, are forcing a critical re-evaluation of the risk-benefit profile of these increasingly popular medications. **GLP-1s** are undeniably changing the landscape of metabolic health, but understanding and mitigating the emerging safety concerns is paramount.
Beyond the Headlines: Understanding the Pancreatitis Link
The connection between GLP-1 receptor agonists and pancreatitis isn’t entirely new. While clinical trials initially showed no significant increase in risk, the sheer scale of real-world usage is now revealing a more nuanced picture. The recent case reports, like the one detailed in Cureus, highlight the potential for acute pancreatitis even in patients without a prior history of the condition. The exact mechanism remains unclear, but theories suggest GLP-1s may exacerbate pre-existing pancreatic inflammation or directly impact pancreatic cell function.
Why the Increased Reporting Now?
Several factors contribute to the increased reporting of pancreatitis cases. First, the dramatic surge in prescriptions means more people are exposed to the potential risk. Second, heightened awareness among both physicians and patients is leading to more thorough investigation of abdominal pain. Finally, the speed at which these drugs induce weight loss may be a contributing factor, potentially stressing the pancreas as it adjusts to altered metabolic demands.
The Emerging Landscape: Beyond Semaglutide and Liraglutide
The GLP-1 class is rapidly evolving. New formulations, including oral semaglutide and combination therapies, are entering the market. Furthermore, research is expanding beyond diabetes and obesity to explore potential applications in cardiovascular disease, neurodegenerative disorders, and even addiction. This broadening scope necessitates a more comprehensive understanding of long-term safety profiles.
The Rise of Dual-Action and Triple-Action Agonists
The next wave of metabolic drugs won’t just target GLP-1. Pharmaceutical companies are developing “twincretins” – drugs that simultaneously activate GLP-1 and GIP receptors – and even “tricretins” adding a third receptor target. These multi-action agonists promise even greater efficacy, but also introduce new layers of complexity regarding potential side effects. Will these more potent drugs amplify the risk of pancreatitis, or will they offer improved safety profiles through different mechanisms of action? This is a critical question that ongoing clinical trials must address.
Predictive Analytics and Personalized Risk Assessment
The future of GLP-1 therapy lies in personalized medicine. Rather than a one-size-fits-all approach, we can anticipate a shift towards predictive analytics that identify individuals at higher risk of adverse events, including pancreatitis. This will involve integrating genetic data, medical history, lifestyle factors, and potentially even biomarkers to create individualized risk scores. Artificial intelligence and machine learning will play a crucial role in analyzing these complex datasets and providing clinicians with actionable insights.
The Role of Continuous Glucose Monitoring (CGM)
CGM technology, already widely used by individuals with diabetes, could also become a valuable tool for monitoring GLP-1 safety. Subtle changes in glucose variability or pancreatic function, detectable through CGM data, might serve as early warning signs of developing pancreatitis. Integrating CGM data with other risk factors could further refine personalized risk assessments.
| Metric | Current Status (2024) | Projected Status (2028) |
|---|---|---|
| GLP-1 Prescription Volume (US) | 40 Million | 120 Million |
| Pancreatitis Reporting Rate (per 10,000 patients) | 0.5 | 1.2 (with increased awareness) |
| Adoption of Personalized Risk Assessment Tools | 5% | 60% |
Frequently Asked Questions About GLP-1s and Pancreatitis
What are the early warning signs of pancreatitis while taking GLP-1s?
Severe abdominal pain that radiates to the back, nausea, vomiting, and fever are all potential symptoms. It’s crucial to contact your doctor immediately if you experience any of these symptoms.
Will the FDA take action regarding the pancreatitis risk?
The FDA is actively monitoring the situation and may require additional safety studies or updates to drug labeling. Increased post-market surveillance is also likely.
Are there alternative weight loss medications with a lower risk of pancreatitis?
Several other weight loss options are available, including lifestyle modifications, other medications, and bariatric surgery. The best choice depends on individual circumstances and risk factors.
The GLP-1 revolution is transforming the treatment of metabolic diseases, but it’s a revolution that demands vigilance. By embracing personalized medicine, leveraging predictive analytics, and prioritizing patient safety, we can harness the immense potential of these drugs while minimizing the risks. The future of weight loss isn’t just about achieving a lower number on the scale; it’s about achieving optimal health and well-being for all.
What are your predictions for the future of GLP-1s and their impact on healthcare? Share your insights in the comments below!
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.