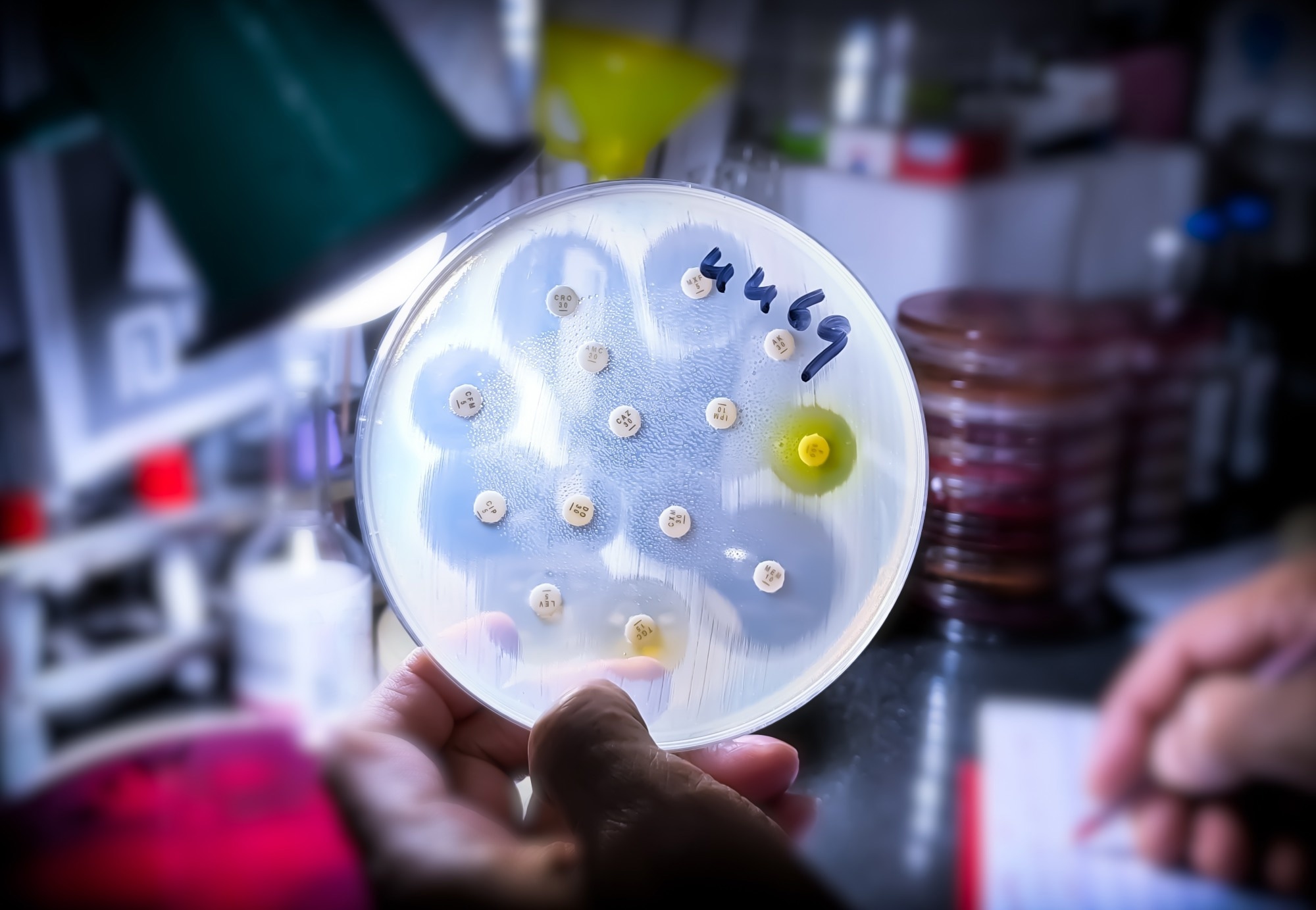
The modern medical establishment is currently locked in a high-stakes arms race against an invisible enemy. With antimicrobial resistance (AMR) contributing to nearly 5 million deaths globally in a single year, the world is facing a “discovery void”—a terrifying gap where bacterial evolution is outstripping our ability to develop new chemical weapons to stop them. For decades, the pharmaceutical industry largely abandoned the dirt beneath our feet, believing the “Golden Age” of soil-derived antibiotics had been exhausted. They were wrong.
- The Pipeline Crisis: Between 2017 and 2023, only 13 new antibacterials were approved, with only two meeting strict WHO innovation criteria for novelty.
- Breaking the “Culturability Barrier”: New technologies like the iChip and metagenomics are allowing scientists to access the 99% of soil microbes that previously refused to grow in laboratory settings.
- The Dual Nature of Soil: While soil is a goldmine for new drug leads (e.g., Teixobactin), it also acts as a global reservoir for resistance genes, necessitating a “One Health” approach to environmental management.
The Discovery Void: Why the “Golden Age” Ended
To understand why the return to soil is so significant, one must understand why we left it. In the mid-20th century, the “Waksman platform”—the systematic culturing of soil microbes—gave us tetracyclines and aminoglycosides. However, by the late 1960s, discovery rates plummeted. This wasn’t because the soil had run out of secrets, but because of the “re-isolation problem.” Scientists kept finding the same common microbes and the same known compounds.
This stagnation was exacerbated by the “culturability crisis.” It is now understood that roughly 99% of environmental microorganisms are unculturable under standard lab conditions; they require specific ecological signals and nutrients that a petri dish simply cannot provide. This technical wall drove the industry toward synthetic drug development, which, while sophisticated, often failed to penetrate the complex outer membranes of Gram-negative bacteria.
The New Frontier: Digital Mining and In Situ Cultivation
We are now entering a second Golden Age, driven by a shift from phenotypic screening (growing things to see if they kill bacteria) to genotype-driven discovery. The integration of three distinct technologies is changing the game:
- Metagenomics & Genome Mining: Scientists can now sequence “microbial dark matter” directly from soil samples, identifying biosynthetic gene clusters (BGCs) without ever needing to grow the microbe in a lab.
- The iChip: By placing microbes in semipermeable membranes and returning them to their natural soil environment, the isolation chip (iChip) allows slow-growing organisms to thrive, leading to the discovery of potent new leads like Teixobactin.
- AI-Assisted Screening: Artificial intelligence is now being used to screen millions of virtual molecules, prioritizing which chemical scaffolds are most likely to be effective before they are ever synthesized in a lab.
These advancements have already yielded promising results. Teixobactin, isolated via the iChip, binds to highly conserved lipid precursors, making it difficult for bacteria to develop resistance. Similarly, lariocidin, a lasso peptide, targets the bacterial ribosome with high efficacy against Acinetobacter baumannii, a pathogen notorious for its resistance.
The Paradox: Soil as Both Cure and Carrier
From a “One Health” perspective, soil is a double-edged sword. While it provides the blueprints for our next generation of antibiotics, it is also a massive reservoir of antimicrobial resistance genes (ARGs). Agricultural runoff, sewage sludge, and manure introduce antibiotic residues into the earth, creating a selective pressure that encourages the evolution of resistance.
This means that the search for new drugs cannot happen in a vacuum. The sustainability of future antibiotic therapy depends on environmental stewardship—managing how we treat our land to ensure we aren’t accelerating the evolution of the very resistance we are trying to cure.
Forward Look: What Happens Next?
The trajectory of antibiotic discovery is moving away from “serendipitous discovery” and toward “predictive engineering.” We should expect the following shifts over the next five to ten years:
1. The Rise of “Synthetic Natural Products”: Rather than simply extracting molecules from soil, researchers will use genome-mining data to “print” optimized versions of these molecules, improving their pharmacokinetic profiles and reducing toxicity.
2. Targeted Gram-Negative Breakthroughs: The focus will shift heavily toward overcoming the outer membrane of Gram-negative pathogens, using AI to design molecules that can bypass efflux pumps—the biological “sump pumps” bacteria use to eject antibiotics.
3. Integrated Soil Monitoring: We will likely see the implementation of global “resistome maps,” where soil monitoring becomes a standard part of public health surveillance to predict the emergence of new resistant strains before they jump from the environment to the clinic.
Ultimately, the battle against AMR will not be won by a single “silver bullet” drug, but by a systemic integration of AI, metagenomics, and ecological management. The dirt beneath our feet is no longer just earth; it is the world’s largest, most complex pharmaceutical library.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.