58
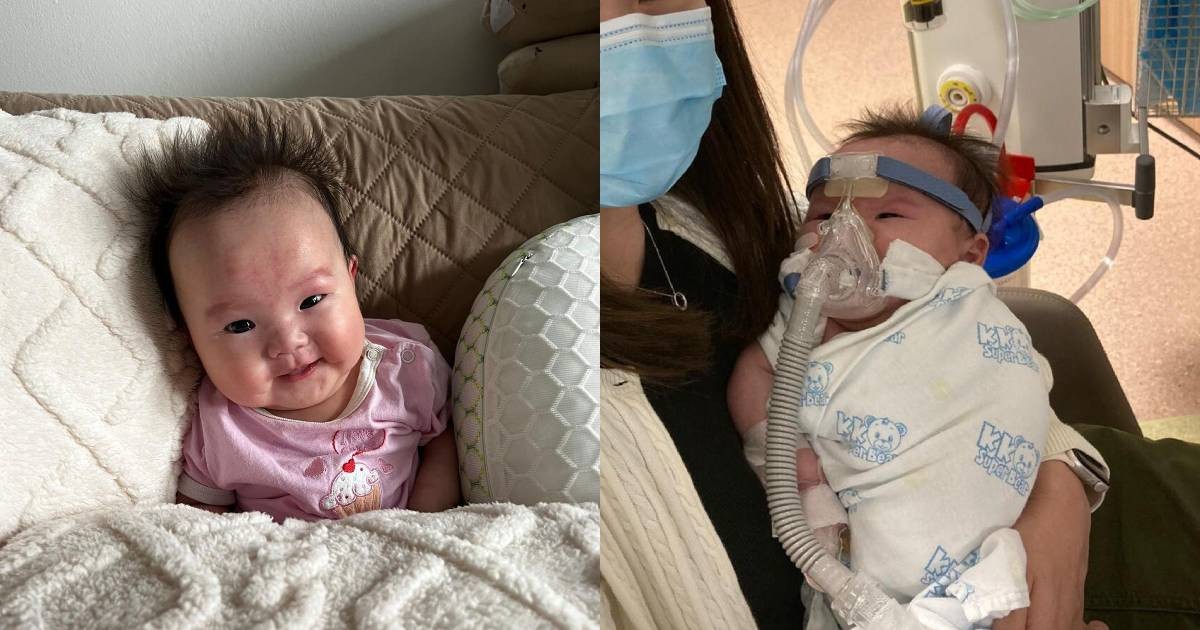
<p>Nearly 80% of rare diseases are genetic in origin, impacting an estimated 300 million people worldwide. But for families like Ginny’s in Singapore, facing a diagnosis of Spinal Muscular Atrophy (SMA), the fight for life extends beyond medical intervention – it’s a desperate race against a multi-million dollar price tag. The recent public appeal for S$2.4 million (approximately $1.78 million USD) to fund gene therapy for 5-month-old Ginny underscores a growing crisis: how do we make potentially curative, yet astronomically expensive, treatments accessible to those who need them most?</p>
<h2>The Escalating Costs of Genetic Cures</h2>
<p>Baby Ginny’s case isn’t isolated. The first FDA-approved gene therapy, Zolgensma, for SMA, carries a list price of $2.125 million. While offering a potential one-time cure, this cost is prohibitive for most families and healthcare systems. This raises a fundamental question: is a life-saving treatment truly accessible if only the wealthiest can afford it? The current landscape is characterized by a complex interplay of pharmaceutical innovation, limited competition, and the inherent challenges of manufacturing these highly personalized therapies.</p>
<h3>Beyond Zolgensma: A Pipeline of Pricey Promises</h3>
<p>Zolgensma was just the beginning. A robust pipeline of gene therapies targeting a wide range of genetic disorders – from hemophilia to inherited blindness – is rapidly advancing. Each new therapy brings with it the potential for dramatic improvements in patient outcomes, but also the specter of similarly exorbitant costs. This isn’t simply a matter of pharmaceutical greed; the research, development, and manufacturing of gene therapies are incredibly complex and expensive. However, the current pricing model is unsustainable and demands innovative solutions.</p>
<h2>Emerging Payment Models: Towards Greater Accessibility</h2>
<p>The high cost of gene therapy is driving exploration of alternative payment models. Several approaches are gaining traction, aiming to spread the financial burden and incentivize value-based care. These include:</p>
<ul>
<li><strong>Outcome-Based Contracts:</strong> Pharmaceutical companies are paid only if the therapy delivers the promised clinical benefit.</li>
<li><strong>Installment Payments:</strong> Spreading the cost over several years, making it more manageable for healthcare systems.</li>
<li><strong>Government Subsidies & Public Funding:</strong> Direct financial assistance to patients or healthcare providers.</li>
<li><strong>Lottery Systems:</strong> While ethically complex, some propose lotteries to allocate limited, expensive therapies.</li>
<li><strong>Risk-Sharing Agreements:</strong> Insurance companies and manufacturers share the financial risk based on treatment outcomes.</li>
</ul>
<p>These models aren’t without their challenges. Determining “success” in outcome-based contracts can be difficult, and long-term follow-up is crucial. However, they represent a critical shift towards a more equitable and sustainable approach to gene therapy financing.</p>
<h3>The Role of Biosimilars and Competition</h3>
<p>As patents on initial gene therapies expire, the emergence of biosimilars – similar, but not identical, versions of the original drug – could drive down prices. However, the complexity of manufacturing gene therapies presents significant hurdles to biosimilar development. Furthermore, regulatory pathways for biosimilar approval are still evolving, potentially delaying market entry and limiting competition. **Gene editing technologies**, like CRISPR, also hold promise for reducing manufacturing costs and increasing accessibility, but are still in early stages of development.</p>
<h2>The Future of Gene Therapy: Beyond Cost</h2>
<p>The conversation around gene therapy extends beyond cost. Long-term safety and efficacy remain paramount concerns. While initial results are promising, the long-term effects of altering a patient’s genome are still largely unknown. Furthermore, equitable access to gene therapy isn’t just about affordability; it’s also about ensuring that clinical trials are representative of diverse populations and that therapies are developed for a wide range of genetic disorders, not just those affecting large, affluent markets.</p>
<table>
<thead>
<tr>
<th>Gene Therapy</th>
<th>Target Disease</th>
<th>Approximate Cost (USD)</th>
</tr>
</thead>
<tbody>
<tr>
<td>Zolgensma</td>
<td>Spinal Muscular Atrophy (SMA)</td>
<td>$2.125 Million</td>
</tr>
<tr>
<td>Luxturna</td>
<td>Inherited Retinal Disease</td>
<td>$850,000 (per eye)</td>
</tr>
<tr>
<td>Glybera (Withdrawn)</td>
<td>Lipoprotein Lipase Deficiency</td>
<td>$1 Million</td>
</tr>
</tbody>
</table>
<p>The story of baby Ginny is a poignant reminder of the immense potential – and the significant challenges – of gene therapy. As these life-changing treatments become more prevalent, we must proactively address the ethical, financial, and logistical hurdles to ensure that they benefit all who need them, not just a privileged few.</p>
<h2>Frequently Asked Questions About Gene Therapy</h2>
<h3>What is the biggest obstacle to wider gene therapy adoption?</h3>
<p>The primary obstacle is cost. The extremely high price tags of current gene therapies make them inaccessible to most patients and healthcare systems, necessitating innovative financing models.</p>
<h3>Will gene editing technologies like CRISPR lower the cost of gene therapy?</h3>
<p>Potentially, yes. CRISPR and other gene editing tools offer the possibility of more efficient and cost-effective manufacturing processes, but these technologies are still under development and require further research.</p>
<h3>Are there any ethical concerns surrounding gene therapy?</h3>
<p>Yes. Concerns include the potential for off-target effects (unintended genetic modifications), equitable access, and the long-term consequences of altering the human genome. Careful regulation and ethical oversight are crucial.</p>
<h3>What role do governments play in making gene therapy more accessible?</h3>
<p>Governments can play a vital role through funding research, negotiating drug prices, providing subsidies, and establishing clear regulatory pathways for gene therapy development and approval.</p>
<p>What are your predictions for the future of gene therapy and its impact on healthcare? Share your insights in the comments below!</p>
<script>
{
"@context": "https://schema.org",
"@type": "NewsArticle",
"headline": "The $2.4 Million Question: Gene Therapy Costs and the Future of Rare Disease Treatment",
"datePublished": "2025-06-24T09:06:26Z",
"dateModified": "2025-06-24T09:06:26Z",
"author": {
"@type": "Person",
"name": "Archyworldys Staff"
},
"publisher": {
"@type": "Organization",
"name": "Archyworldys",
"url": "https://www.archyworldys.com"
},
"description": "The case of baby Ginny highlights the soaring costs of gene therapy. We explore the financial barriers, emerging payment models, and the potential for wider access to these life-changing treatments."
}
{
"@context": "https://schema.org",
"@type": "FAQPage",
"mainEntity": [
{
"@type": "Question",
"name": "What is the biggest obstacle to wider gene therapy adoption?",
"acceptedAnswer": {
"@type": "Answer",
"text": "The primary obstacle is cost. The extremely high price tags of current gene therapies make them inaccessible to most patients and healthcare systems, necessitating innovative financing models."
}
},
{
"@type": "Question",
"name": "Will gene editing technologies like CRISPR lower the cost of gene therapy?",
"acceptedAnswer": {
"@type": "Answer",
"text": "Potentially, yes. CRISPR and other gene editing tools offer the possibility of more efficient and cost-effective manufacturing processes, but these technologies are still under development and require further research."
}
},
{
"@type": "Question",
"name": "Are there any ethical concerns surrounding gene therapy?",
"acceptedAnswer": {
"@type": "Answer",
"text": "Yes. Concerns include the potential for off-target effects (unintended genetic modifications), equitable access, and the long-term consequences of altering the human genome. Careful regulation and ethical oversight are crucial."
}
},
{
"@type": "Question",
"name": "What role do governments play in making gene therapy more accessible?",
"acceptedAnswer": {
"@type": "Answer",
"text": "Governments can play a vital role through funding research, negotiating drug prices, providing subsidies, and establishing clear regulatory pathways for gene therapy development and approval."
}
}
]
}
</script>Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.