Understanding how bacteria divide isn’t just an academic exercise; it’s a critical step in combating antibiotic resistance. As bacteria evolve to evade existing drugs, pinpointing the fundamental mechanisms of their life cycle – like cell division – provides new targets for intervention. A team at the Universitat Autònoma de Barcelona (UAB) has now delivered a significant breakthrough, revealing the molecular details of how bacteria regulate this essential process, potentially opening doors to a new generation of antibacterial strategies.
- Molecular Mechanism Revealed: Researchers have identified how the MraZ protein interacts with the dcw gene cluster to control bacterial cell division.
- Structural Change is Key: The MraZ protein undergoes a surprising structural deformation – essentially breaking its “donut” shape – to bind to DNA and activate cell division.
- Broad Implications: This regulatory system appears to be universal across most bacterial species, suggesting a wide-ranging impact for future research.
Cell division is, of course, fundamental to all life. In bacteria, this process is orchestrated by a set of genes known as the dcw operon, responsible for building both the cell wall and the machinery for division. The dcw operon doesn’t activate itself; it requires transcription factors – proteins that bind to DNA and initiate the process of reading the genetic code. MraZ is a key player here, acting as the primary regulator of the dcw operon in most bacterial species. Previous understanding of *how* MraZ did this was largely based on inference, relying on biochemical experiments and computer models. This new research moves beyond that.
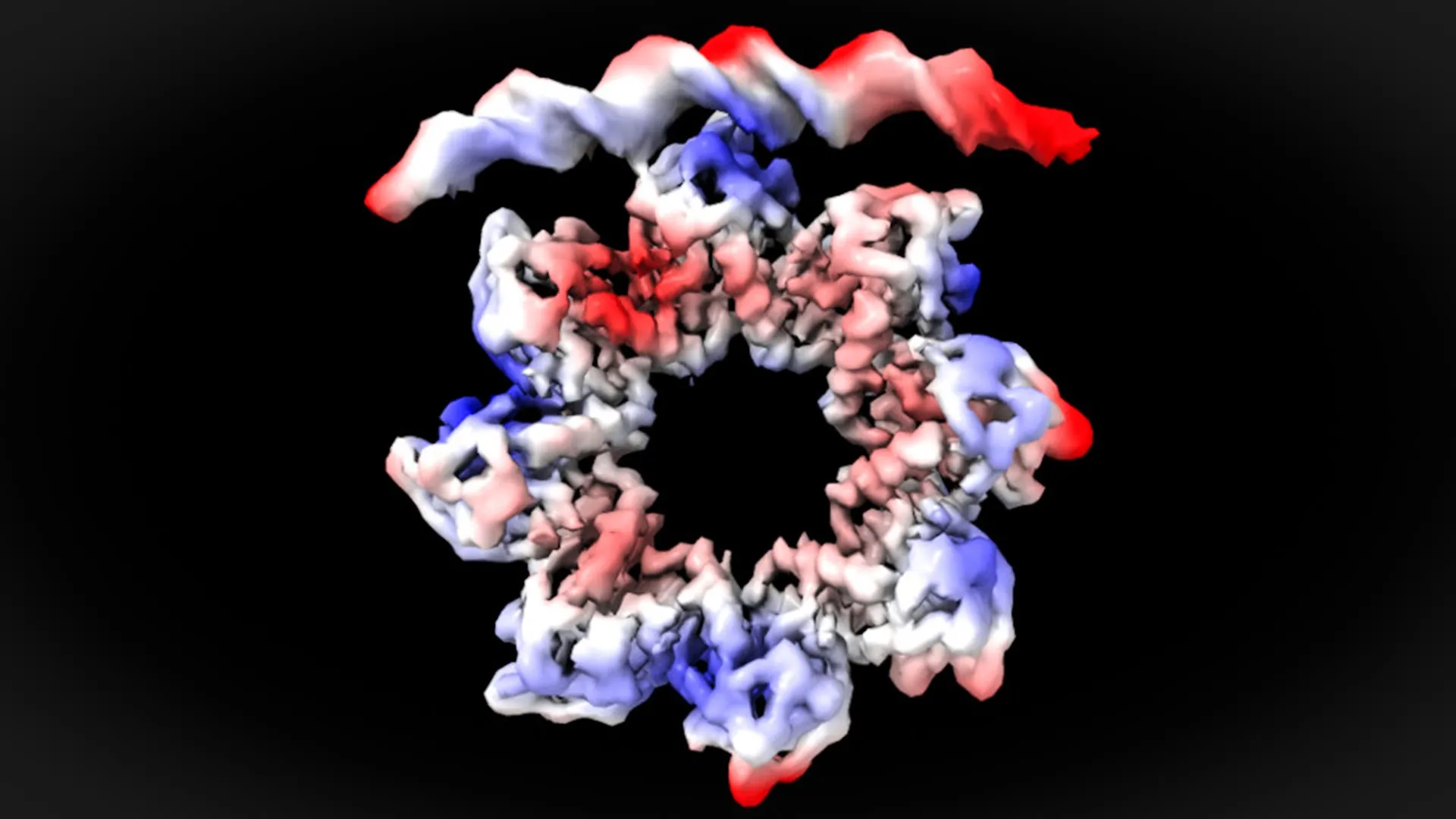
The UAB team, led by David Reverter, utilized advanced structural biology techniques – X-ray crystallography and cryo-electron microscopy – to visualize the interaction between MraZ and the dcw operon’s promoter region (the DNA sequence where transcription begins) at near-atomic resolution. They focused on Mycoplasma genitalium, a bacterium with a remarkably small genome, making it an ideal model for this type of detailed study. What they discovered was unexpected. The promoter region contains four repeating DNA sequences, and MraZ, normally shaped like a donut, must dramatically alter its structure to effectively bind to these sequences. The donut essentially breaks apart, allowing portions of the protein to attach to the DNA.
This isn’t just a fascinating structural observation; it’s a fundamental shift in our understanding of bacterial cell division. The ability to directly visualize this interaction provides a concrete foundation for future research and drug development.
The Forward Look
The most immediate impact will be a surge in research focused on disrupting the MraZ-dcw operon interaction. Now that we understand the precise mechanism, scientists can begin designing molecules that specifically target this process, preventing bacteria from dividing. Expect to see increased investment in structure-based drug design, leveraging the atomic-level data provided by Reverter’s team. Furthermore, the universality of this mechanism across bacterial species is particularly encouraging. A successful drug targeting MraZ could potentially be effective against a broad spectrum of bacterial infections. However, the speed at which bacteria evolve resistance means this is a race against time. The next few years will be critical in translating this fundamental discovery into tangible clinical solutions. We can also anticipate research exploring whether similar structural changes occur in other bacterial regulatory proteins, potentially revealing a common theme in gene regulation.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.