The intricate relationship between the gut microbiome, immune function, and brain health is coming into sharper focus, thanks to a comprehensive study published today. Researchers at the University of Pennsylvania, the Arc Institute, and Stanford University have demonstrated, in mice, that age-related cognitive decline is significantly influenced by shifts in gut microbial composition and associated metabolic changes. This isn’t simply a correlation; the study provides compelling evidence of a causal link, opening new avenues for preventative and therapeutic interventions targeting age-related neurodegeneration. The findings build on a growing body of research highlighting the gut-brain axis, but crucially, this work identifies specific microbial players and pathways involved in cognitive function, and importantly, demonstrates a degree of reversibility.
Key Takeaways
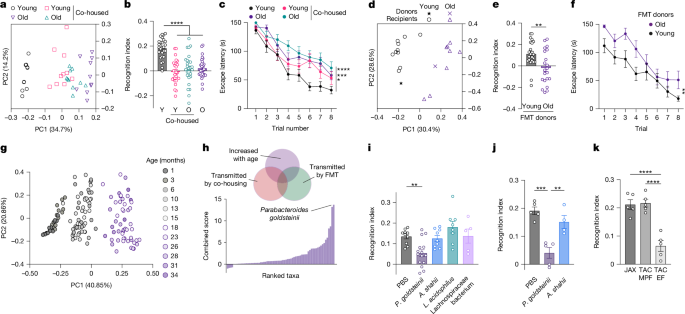
- Microbiome Shifts Drive Cognitive Decline: Age-related changes in gut bacteria, specifically a decrease in Parabacteroides goldsteinii and Alistipes shahii, are linked to impaired cognitive function in mice.
- Metabolic Pathways are Key: The study identifies specific microbial metabolites, including short-chain fatty acids, that mediate the gut-brain connection and influence neuronal activity.
- Potential for Intervention: Fecal microbiota transplantation (FMT) and targeted dietary interventions show promise in restoring cognitive function, suggesting a path towards preventative strategies for age-related cognitive decline.
The Deep Dive: Unraveling the Gut-Brain Connection
For years, scientists have suspected a strong connection between the gut microbiome and brain health. This study provides a mechanistic understanding of *how* this connection operates. Researchers utilized a multi-faceted approach, including germ-free mice, fecal microbiota transplantation, antibiotic treatments, and detailed analysis of microbial composition, metabolites, and brain activity. A key finding was the age-related decline in specific bacterial species, notably P. goldsteinii and A. shahii. These bacteria are associated with the production of metabolites that appear to be crucial for maintaining neuronal health and synaptic plasticity.
The researchers meticulously demonstrated that transferring the gut microbiome from old mice to young, germ-free mice induced cognitive deficits in the young recipients. Conversely, transplanting the microbiome from young mice into old mice partially restored cognitive function. This bidirectional transfer of cognitive phenotypes strongly suggests a causal role for the gut microbiome. Further investigation revealed that specific metabolites produced by these bacteria, particularly short-chain fatty acids (SCFAs), influence neuronal activity in the nodose ganglia – a critical relay station for vagal nerve signaling – and ultimately impact cognitive performance. The study also identified GPR84, a receptor expressed on myeloid cells, as a key mediator of this gut-brain communication. Activation of GPR84 by microbial metabolites appears to modulate immune responses in the gut and influence brain function.
Interestingly, the study also explored the role of phages – viruses that infect bacteria – in shaping the gut microbiome. Targeting specific phages to reduce levels of detrimental bacteria showed some promise in improving cognitive outcomes, highlighting a potential novel therapeutic strategy.
The Forward Look: Implications and Future Research
This research has significant implications for how we approach age-related cognitive decline. While the study was conducted in mice, the identified microbial players and metabolic pathways are conserved in humans, suggesting that similar mechanisms may be at play. The most immediate next step is to validate these findings in human cohorts. Large-scale studies correlating gut microbiome composition with cognitive performance in aging populations are needed.
However, the potential for intervention is particularly exciting. The study suggests several avenues for therapeutic development:
- Probiotic and Prebiotic Strategies: Developing targeted probiotics or prebiotics to promote the growth of beneficial bacteria like P. goldsteinii and A. shahii could be a preventative measure against cognitive decline.
- Dietary Interventions: Identifying dietary patterns that support a healthy gut microbiome could be a powerful tool for maintaining cognitive function. The study points to the importance of fiber-rich diets that promote SCFA production.
- Phage Therapy: Developing phage-based therapies to selectively target detrimental bacteria in the gut could offer a more precise approach to microbiome modulation.
- GPR84 Modulation: Investigating compounds that can modulate GPR84 activity could offer a novel therapeutic target for improving gut-brain communication.
Legal experts anticipate increased investment in microbiome-focused biotech companies and a surge in clinical trials exploring the therapeutic potential of gut microbiome modulation for neurodegenerative diseases. The FDA is likely to face increasing pressure to establish clear regulatory pathways for microbiome-based therapies. Furthermore, this research underscores the importance of personalized medicine, as individual gut microbiome compositions may influence susceptibility to cognitive decline and response to therapeutic interventions. The era of treating the brain through the gut is rapidly approaching, and this study provides a crucial roadmap for navigating this exciting new frontier.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.