New Treatment Approach Significantly Improves Outcomes for Multiple Myeloma Patients
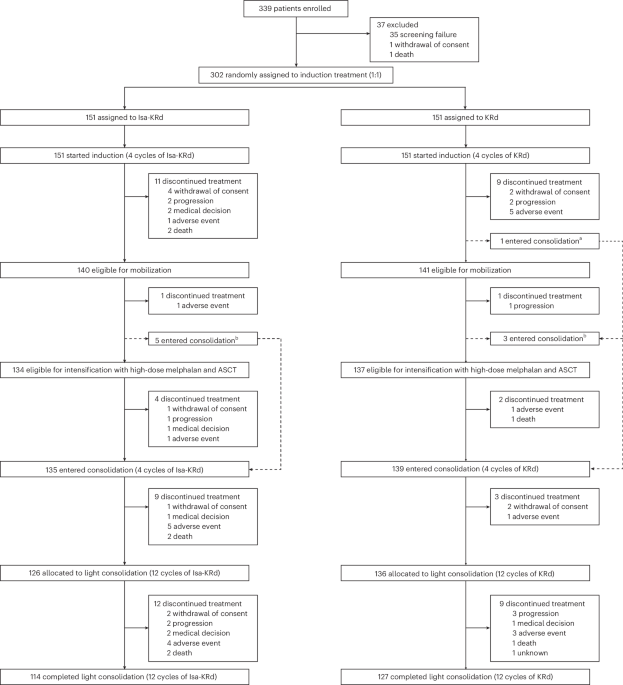
Groundbreaking results from the phase 3 IsKia trial reveal a substantial improvement in treatment efficacy for individuals newly diagnosed with multiple myeloma who are eligible for stem cell transplantation. Patients receiving isatuximab in combination with carfilzomib, lenalidomide, and dexamethasone before and after transplant demonstrated significantly higher rates of achieving measurable residual disease negativity – a key indicator of treatment success – compared to those receiving carfilzomib, lenalidomide, and dexamethasone alone. This advancement offers renewed hope for a more durable remission and potentially longer survival for those battling this challenging blood cancer.
Understanding Multiple Myeloma and the Role of Measurable Residual Disease
Multiple myeloma is a cancer that forms in plasma cells, a type of white blood cell responsible for producing antibodies. While treatments have improved significantly in recent years, the disease often returns, even after initial remission. Measurable residual disease (MRD) refers to the presence of cancer cells remaining in the body at levels undetectable by standard tests. Achieving MRD negativity is increasingly recognized as a critical goal of treatment, as it correlates with improved progression-free survival and overall survival rates.
The standard of care for transplant-eligible patients typically involves induction therapy to reduce the cancer burden, followed by autologous stem cell transplantation, and then consolidation therapy to maintain remission. The IsKia trial investigated whether adding isatuximab, an antibody targeting a protein found on myeloma cells, to this established regimen could enhance outcomes. The results suggest a clear benefit, particularly in achieving deeper remissions as evidenced by higher MRD negativity rates.
Carfilzomib, lenalidomide, and dexamethasone form the backbone of many multiple myeloma treatment plans. The National Cancer Institute provides comprehensive information on these medications and their mechanisms of action. Isatuximab works by binding to CD38, a protein highly expressed on myeloma cells, triggering the body’s immune system to destroy them.
Did You Know?:
The implications of these findings extend beyond simply improving MRD negativity. Researchers believe that deeper remissions achieved with isatuximab may translate into longer-term benefits for patients, potentially delaying disease relapse and improving quality of life. However, further long-term follow-up is needed to confirm these benefits definitively.
What are the potential long-term effects of achieving MRD negativity in multiple myeloma? And how might this new combination therapy impact the future of myeloma treatment protocols?
For more information on clinical trials and ongoing research in multiple myeloma, visit the Multiple Myeloma Research Foundation website.
Frequently Asked Questions About the IsKia Trial and Multiple Myeloma Treatment
Share this article with your network to help raise awareness about advancements in multiple myeloma treatment. Join the conversation and share your thoughts in the comments below.
Disclaimer: This article provides general information and should not be considered medical advice. Please consult with a qualified healthcare professional for any health concerns or before making any decisions related to your treatment.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.