Chronic pain, a condition affecting millions and costing the US economy billions annually, may not be a signaling problem – but an energy problem. New research from Duke University, published in Nature, reveals a surprising mechanism: pain-sensing neurons rely on direct energy transfers from neighboring support cells via microscopic “tunnels.” This isn’t just a fascinating biological discovery; it fundamentally shifts our understanding of chronic pain and opens entirely new therapeutic avenues, moving beyond simply blocking pain signals to addressing the root cause of nerve dysfunction.
- Mitochondrial Hand-Off: Pain neurons receive vital energy (mitochondria) directly from support cells through temporary cellular bridges.
- Disrupted Transfer = Pain: Blocking this energy transfer in mice led to increased pain sensitivity and nerve fiber deterioration, mimicking peripheral neuropathy.
- Therapeutic Potential: Restoring mitochondrial supply, or boosting the transfer process, shows promise in alleviating nerve pain – a potential paradigm shift in treatment.
The Energy Crisis in Your Nerves
For decades, the prevailing view of nerve pain focused on the transmission of pain signals – how quickly and efficiently they travel to the brain. Pharmaceutical interventions largely target these signals, aiming to dampen or block them. However, the efficacy of these treatments is often limited, and many patients experience persistent pain. This new research suggests a critical missing piece: the metabolic health of the neurons themselves. Nerve fibers, particularly the smaller ones responsible for certain types of pain, are incredibly long – some stretching over three feet. Maintaining sufficient energy levels along these distances is a significant challenge. The discovery that support cells, called satellite glial cells, actively supplement neurons with mitochondria – the cell’s powerhouses – explains how these long fibers can function. These aren’t just passive support cells; they’re actively engaged in a vital energy supply chain.
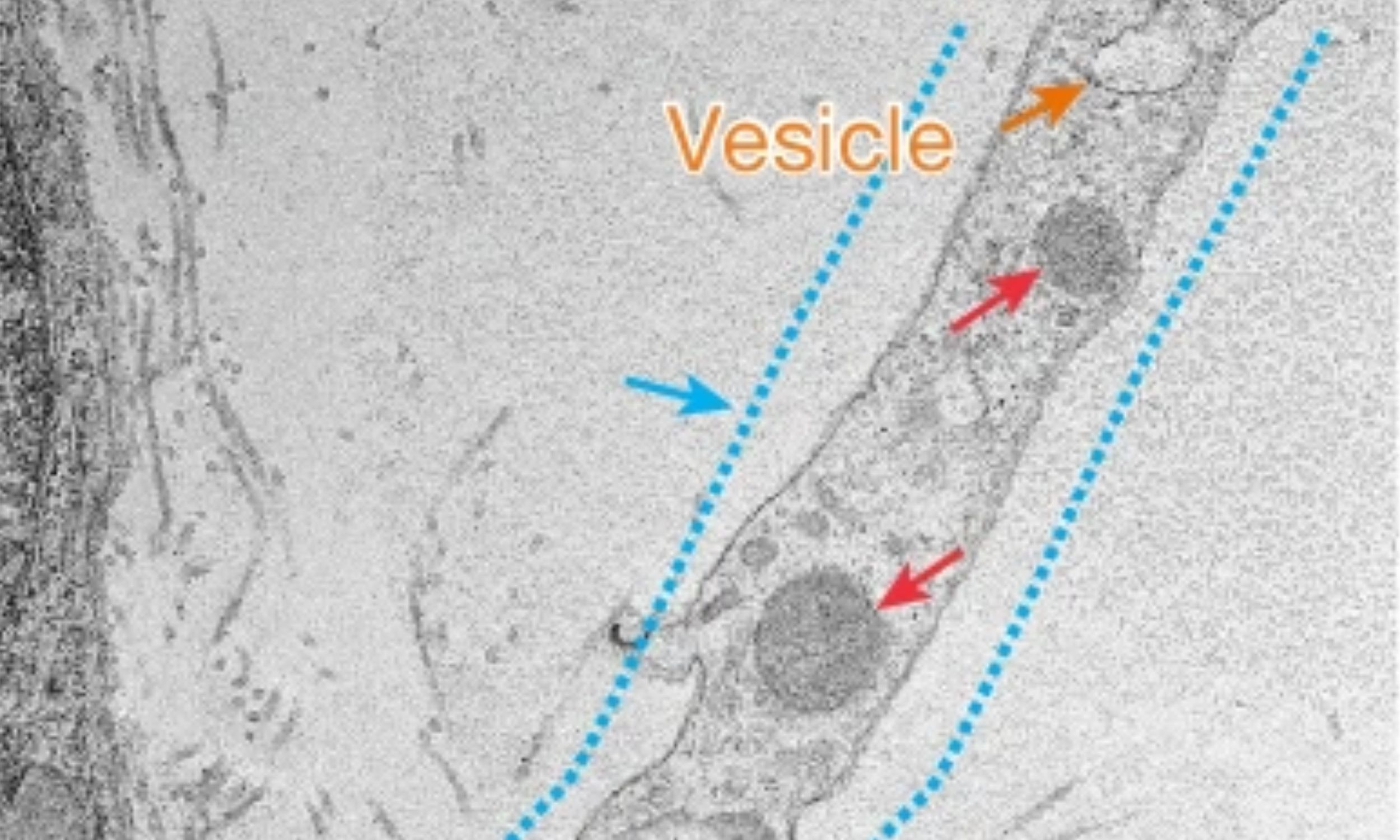
Tunneling Nanotubes: A Surprisingly Efficient Delivery System
The mechanism of this energy transfer is equally remarkable. Satellite glial cells extend temporary, dynamic structures called tunneling nanotubes – essentially microscopic bridges – directly to the surface of the neurons. These nanotubes, built with the help of a protein called MYO10, act as conduits for delivering mitochondria. The transient nature of these tubes – they assemble and disassemble within minutes – highlights the constant, active process of energy support. Importantly, this also reveals a vulnerability: disrupting the formation of these nanotubes, or the activity of MYO10, effectively cuts off the energy supply, leading to nerve dysfunction. The fact that these nanotubes can transport such large organelles (mitochondria) is a significant finding, suggesting they may be used for other forms of cellular communication and cargo delivery.
Looking Ahead: From Mice to Meaningful Therapies
While the research is currently limited to mouse models and donated human tissue, the implications are profound. The observation that mitochondrial transfer is reduced in conditions like diabetes and chemotherapy-induced neuropathy provides a clear link between metabolic dysfunction and chronic pain. The successful demonstration of pain relief in injured mice by delivering healthy glial cells or purified mitochondria is encouraging, but significant hurdles remain. The biggest challenge will be developing a safe and effective method for delivering mitochondria or boosting mitochondrial transfer in humans. Inflammation triggered by injected mitochondria is a major concern. Furthermore, targeting the smaller nerve fibers, which appear to be more vulnerable, will require precise delivery mechanisms.
However, the potential payoff is enormous. Instead of simply masking pain, therapies that address the underlying energy deficit could offer long-term relief and potentially prevent nerve damage. Researchers are already exploring ways to enhance MYO10 activity and stabilize tunneling nanotubes. The next phase of research will likely focus on identifying compounds that can promote mitochondrial biogenesis (the creation of new mitochondria) within neurons, or that can enhance the efficiency of mitochondrial transfer. This discovery reframes chronic pain as a metabolic disease, opening up a completely new frontier in pain management and potentially impacting the treatment of a wide range of neurological disorders. Expect to see a surge in investment and research focused on glial cell biology and mitochondrial function in the coming years.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.