For decades, the medical community viewed neuroinflammation as the “enemy”—a destructive byproduct of diseases like Alzheimer’s and Parkinson’s that needed to be suppressed at all costs. However, a sweeping review published in the Journal of Clinical Investigation reveals a far more nuanced reality: the brain’s immune system is not merely a passenger in neurodegeneration, but a complex regulator that can either stall the disease or accelerate it, depending entirely on timing and context.
- The Paradox of Inflammation: While chronic inflammation drives decay, initial immune responses (via TLRs) are often essential for clearing toxic proteins; suppressing them can actually worsen cognitive decline.
- Disease-Specific T-Cell Roles: The adaptive immune system is not uniform; CD4+ T cells may protect the brain in Alzheimer’s but prove neurotoxic in Parkinson’s.
- The “Priming” Effect: Aging, viral infections, and traumatic brain injuries (TBI) restructure the CNS immune environment, lowering the threshold for neurodegenerative onset.
The Deep Dive: A Double-Edged Sword in the CNS
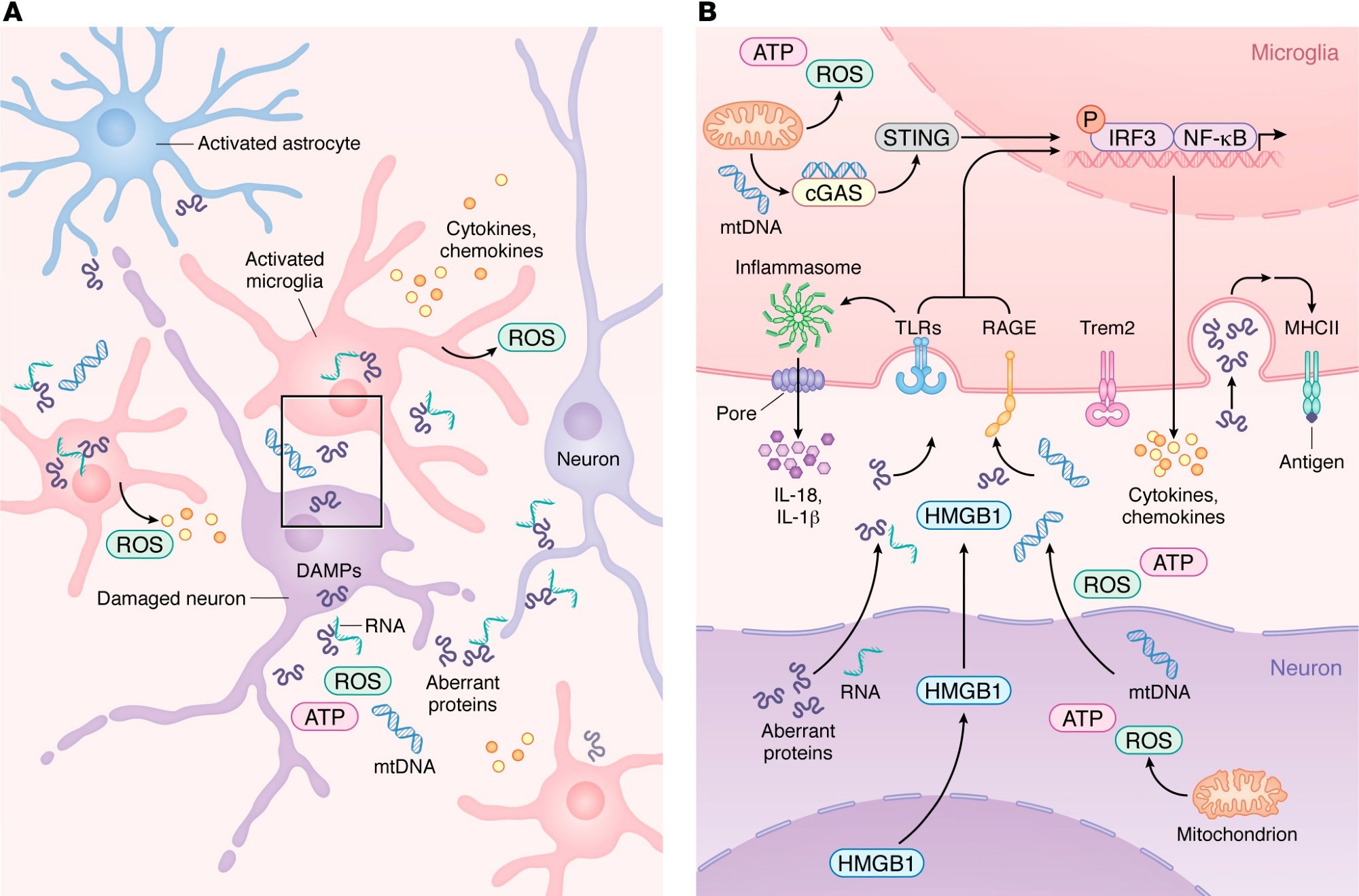
The central nervous system (CNS) is not an “immune-privileged” void but a highly regulated environment. Under normal conditions, resident microglia and astrocytes maintain the blood-brain barrier (BBB) and clear cellular debris. The crisis begins when “danger-associated molecular patterns” (DAMPs)—such as phosphorylated tau and amyloid $beta$—trigger pattern recognition receptors (PRRs) like TLR2 and TLR4.
The critical insight from this research is the temporal shift of these responses. In the early stages of neurodegeneration, microglial activation is a defensive necessity. The review notes that pharmacologically inhibiting certain Toll-like receptors (TLRs) actually increases the burden of amyloid $beta$ and accelerates cognitive loss. This suggests that the brain requires an initial “inflammatory burst” to attempt cleanup. The pathology emerges when this response becomes chronic and maladaptive, shifting from a protective state to a “reactive” state that fuels neuronal death.
Furthermore, the role of the adaptive immune system—specifically T cells—adds a layer of complexity. The discovery that CD4+ T cells are neuroprotective in Alzheimer’s models but pathogenic in Parkinson’s models suggests that we cannot treat “neuroinflammation” as a single entity. The immune system’s behavior is dictated by the specific antigen it is fighting and the local signaling cues of the diseased tissue.
The Forward Look: Toward Precision Immunomodulation
This synthesis of evidence signals a pivot in how future therapies for Alzheimer’s, Parkinson’s, and ALS will be developed. The era of broad-spectrum anti-inflammatories is likely ending, as these drugs risk stripping the brain of its primary defense mechanisms.
What to watch for in the coming years:
- Temporal Mapping: Researchers will likely move toward “stage-dependent” therapies—drugs that support immune activation in the early stages of protein misfolding but suppress it once the disease reaches a chronic, maladaptive phase.
- TREM2-Targeted Therapies: Given that TREM2 polymorphisms are risk factors comparable to the APOE $epsilon$4 allele, expect an increase in clinical trials focusing on modulating TREM2 to steer microglia back toward a homeostatic, protective state.
- T-Cell Engineering: The divergence in T-cell behavior (protective vs. toxic) opens the door for cell-based therapies. We may see the development of antigen-specific T-cell infusions designed to clear specific plaques without triggering a generalized inflammatory storm.
- Preventative “Immune Hygiene”: As the link between viral infections, TBI, and later-life neurodegeneration strengthens, clinical focus may shift toward mitigating CNS inflammation immediately following a head injury or systemic infection to prevent the “priming” of the brain for future decay.
Ultimately, the goal is no longer to silence the brain’s immune system, but to conduct it—ensuring the defense remains a shield rather than becoming a weapon.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.