New Drug Combination Marks Ewing Sarcoma Treatment Breakthrough in Phase 1/2 Trial
In a pivotal shift for pediatric and adolescent oncology, a new therapeutic strategy is showing remarkable promise in treating one of the most challenging bone cancers.
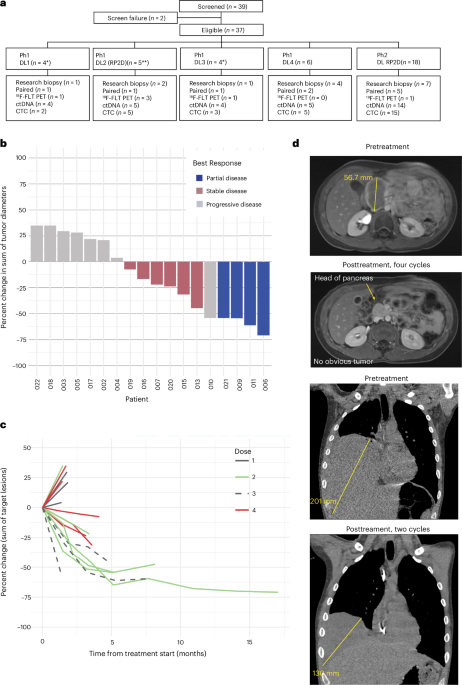
Researchers have unveiled a potent combination therapy targeting the genetic engine of Ewing sarcoma, yielding what experts describe as encouraging clinical response rates in a recent trial.
The study, detailing a Phase 1/2 clinical trial, demonstrates that the combined administration of trabectedin and low-dose irinotecan can effectively stifle the growth of malignant cells by neutralizing a specific transcription factor.
These findings published in Nature Medicine suggest that hitting the cancer’s primary driver—the EWS::FLI1 protein—could redefine the standard of care for patients who have exhausted traditional options.
Could this precision-based approach finally break the plateau in Ewing sarcoma survival rates? Furthermore, how might this combination therapy alter the toxicity profiles currently associated with aggressive chemotherapy?
The Science of the Strike: Understanding EWS::FLI1
To appreciate the magnitude of this Ewing sarcoma treatment breakthrough, one must understand the “master switch” that drives the disease: the EWS::FLI1 fusion protein.
In the vast majority of Ewing sarcoma cases, a genetic translocation creates this abnormal transcription factor. This protein essentially hijacks the cell’s machinery, forcing it to divide uncontrollably while evading the body’s natural immune defenses.
The Synergistic Power of Trabectedin and Irinotecan
For years, the oncology community has sought a way to “turn off” EWS::FLI1, but the protein’s structure made it an elusive target for traditional drugs.
The brilliance of the current trial lies in the synergy between trabectedin and irinotecan. When administered at specific concentrations, these two agents work in tandem to inhibit the activity of the EWS::FLI1 factor more effectively than either drug could alone.
By suppressing this driver, the treatment doesn’t just attack the tumor’s surface; it disrupts the very blueprint the cancer uses to survive and expand.
For more detailed information on the biology of sarcomas, the National Cancer Institute provides comprehensive resources on bone and soft tissue malignancies.
Moving Toward Personalized Oncology
This trial represents a broader move toward “mechanism-based” therapy. Rather than utilizing a “sledgehammer” approach with high-dose systemic chemotherapy, physicians are now focusing on the molecular vulnerabilities of the tumor.
The use of low-dose irinotecan is particularly noteworthy, as it seeks to maximize the inhibitory effect on EWS::FLI1 while minimizing the debilitating side effects often seen in aggressive cancer regimens.
The American Cancer Society emphasizes that such targeted approaches are key to improving the long-term quality of life for cancer survivors.
As this trial moves into subsequent phases, the medical community remains cautiously optimistic that this duo of medications will provide a lifeline to those battling this aggressive malignancy.
Frequently Asked Questions
What is the latest Ewing sarcoma treatment breakthrough?
The latest breakthrough involves a combination of trabectedin and low-dose irinotecan, which targets the EWS::FLI1 transcription factor to improve clinical response rates.
How does the trabectedin and irinotecan combination work?
This combination is designed to inhibit the activity of the EWS::FLI1 transcription factor, which is a primary driver of tumor growth in Ewing sarcoma.
What were the results of the Ewing sarcoma treatment breakthrough trial?
The Phase 1/2 trial reported encouraging clinical response rates among patients receiving the specific concentrations of these two drugs.
Is this Ewing sarcoma treatment breakthrough approved for general use?
The results are from a Phase 1/2 trial, meaning further larger-scale testing is typically required before widespread regulatory approval.
Why is targeting EWS::FLI1 important for Ewing sarcoma treatment?
EWS::FLI1 is the hallmark genetic driver of the disease; successfully inhibiting it strikes at the root of the cancer’s ability to proliferate.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
Join the Conversation: Do you believe targeted genetic therapy is the future of pediatric oncology? Share this article with your network and tell us your thoughts in the comments below.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.