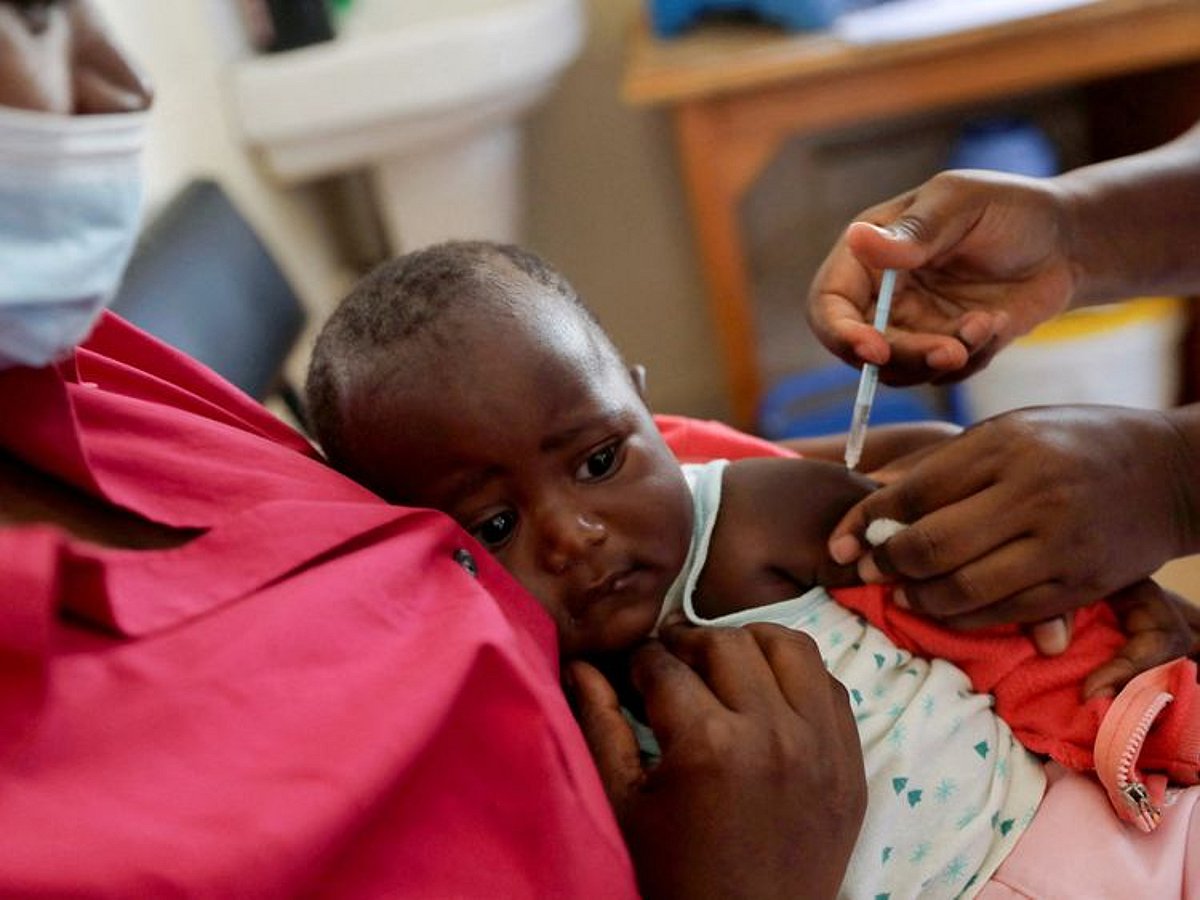
Closing the Vulnerability Gap: How the First WHO-Approved Malaria Treatment for Infants Reshapes Global Neonatal Health
For decades, the most vulnerable patients in the global fight against infectious diseases have been the ones least served by pharmaceutical innovation. Until now, newborns and infants were often treated with “off-label” dosages—essentially adult medications diluted to fit a tiny frame—a practice that is as imprecise as it is risky. The World Health Organization’s (WHO) recent prequalification of the first malaria treatment for infants represents more than just a new drug; it is a fundamental shift in how the global medical community approaches neonatal pharmacology.
The Critical Gap in Pediatric Care
Malaria remains a leading cause of childhood mortality, particularly in sub-Saharan Africa. However, the physiological differences between a newborn and an adult are vast, affecting how drugs are absorbed, metabolized, and excreted.
When treatments are not specifically formulated for infants, the risk of under-dosing—which leads to treatment failure—or over-dosing—which can cause toxicity—is dangerously high. By approving a treatment designed specifically for this age group, the WHO is effectively closing a lethal gap in the healthcare continuum.
Beyond Prequalification: What the Approval Actually Means
WHO prequalification is not merely a regulatory “thumbs up.” It is the gold standard that allows United Nations agencies and international donors to purchase and distribute vaccines and medicines with confidence. This approval triggers a massive logistical chain, moving the treatment from the laboratory into the most remote clinics in the world.
Solving the “Dosing Dilemma”
The primary innovation here is precision. Targeted formulations allow for accurate dosing based on the unique biological needs of an infant, reducing the margin of error for healthcare workers in high-pressure environments. This ensures that the parasite is cleared from the system efficiently without compromising the infant’s developing organs.
Scaling Distribution in Resource-Limited Settings
The challenge now shifts from chemistry to logistics. The success of this treatment depends on “last-mile” delivery. We are likely to see an increase in community health worker training and a push for integrated neonatal care packages that combine this treatment with existing prenatal and postnatal services.
The Ripple Effect: A Blueprint for Future Pediatric Therapeutics
This milestone serves as a proof-of-concept for the pharmaceutical industry. It signals that there is a viable, necessary market for targeted pediatric medicines, challenging the industry to stop treating children as “small adults.”
If this model of targeted formulation succeeds for malaria, we can expect a similar wave of innovation for other neonatal killers, such as pneumonia and neonatal sepsis. We are entering an era of precision pediatrics, where the drug is as specific as the patient.
| Feature | Traditional Approach | New Targeted Treatment |
|---|---|---|
| Formulation | Diluted adult medications | Age-specific pediatric formulation |
| Dosing Accuracy | High risk of variance/error | High precision based on infant weight |
| Safety Profile | Off-label; higher toxicity risk | Clinically validated for newborns |
Challenges Remaining on the Path to Eradication
While the medicine exists, the disease persists. The rollout of this treatment must occur alongside systemic improvements. Malaria eradication cannot rely on a pill alone; it requires the simultaneous deployment of insecticide-treated nets, the scaling of malaria vaccines, and the strengthening of fragile health systems.
Furthermore, the global community must ensure that the cost of these targeted formulations remains affordable. The true test of this breakthrough will not be its efficacy in a trial, but its accessibility in a rural village in the Congo or South Sudan.
Frequently Asked Questions About Malaria Treatment for Infants
How does the new treatment differ from previous options?
Previous options were often adapted adult versions. The new treatment is specifically formulated for the physiological needs of newborns and infants, ensuring safer and more effective dosing.
What is the role of WHO prequalification?
Prequalification is a rigorous verification process that ensures a medicine meets global standards of quality, safety, and efficacy, enabling international procurement agencies to distribute it globally.
Will this lead to the total eradication of malaria?
While it significantly reduces infant mortality, eradication requires a multi-pronged approach including vaccines, vector control, and systemic healthcare investment.
When will this treatment be available in all affected regions?
Availability depends on national procurement and logistics, but WHO prequalification typically accelerates the rollout through international health partnerships.
The approval of a dedicated malaria treatment for infants is a victory for health equity. It acknowledges that the smallest patients deserve the most precise care. By moving toward targeted neonatal medicine, the world is not just fighting a parasite; it is redefining the standard of care for the next generation. The goal is no longer just survival, but a healthy start for every child, regardless of where they are born.
What are your predictions for the future of pediatric pharmacology? Do you believe targeted treatments will accelerate the goal of malaria eradication? Share your insights in the comments below!
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.