The “Wild West” era of off-label cancer treatment—where clinicians prescribe non-approved drugs based on scientific intuition and hope—is facing a necessary reckoning. For years, patients with exhausted treatment options have navigated a precarious landscape of high-cost, high-toxicity therapies with little to no systematic oversight. However, the long-term results of the Drug Rediscovery Protocol (DRUP) provide a critical blueprint for transitioning from desperate gambling to structured, precision-guided medicine.
- The Precision Paradox: While 34.9% of patients saw clinical benefit, the high rate of severe toxicities (28.4% Grade 3+) underscores the danger of off-label use without rigorous trial oversight.
- Histology Still Matters: Despite the push for “tumor-agnostic” treatments, tissue context remained a decisive factor in nearly a quarter of drug-target subgroups, proving that a mutation alone is not always a sufficient roadmap.
- The Patent Barrier: Commercial interests often kill medical progress; patent expiry frequently discouraged pharmaceutical companies from expanding promising cohorts, leaving viable treatments inaccessible.
The Deep Dive: Moving Beyond the “Mutation Match”
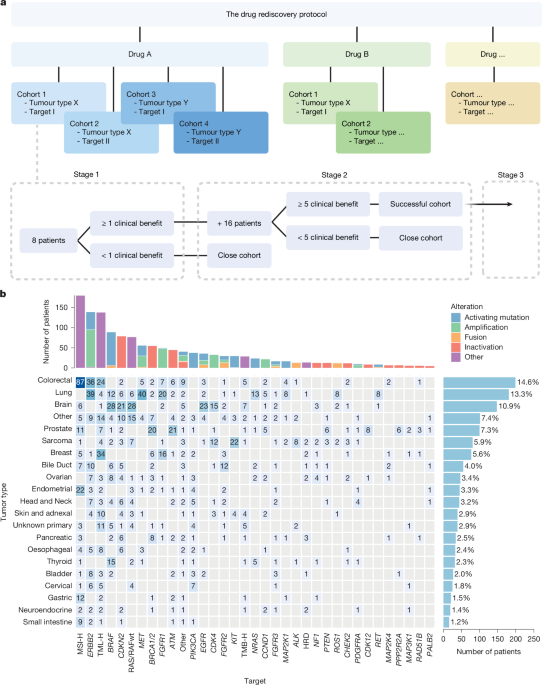
The core ambition of precision oncology has been to treat the mutation, not the organ. The DRUP study, spanning nine years and over 1,600 patients, reveals that while this “tumor-agnostic” approach can produce “exceptional responders”—such as those with BRAF p.V600E-mutated brain tumors—it is not a universal solvent. The data suggests that the most successful interventions are those grounded in high-evidence frameworks like ESCAT or OncoKB, rather than mere biological plausibility.
One of the most sobering findings is the gap between clinician optimism and clinical reality. The study noted high interest in CDK4/6 inhibitors, yet efficacy proved limited. This highlights a systemic risk in oncology: “hope-driven” prescribing. When clinicians pursue targets that look good on paper but fail in practice, patients are exposed to unnecessary toxicity and financial strain without a corresponding increase in survival.
Furthermore, the study exposes a regulatory flaw in how we define “success.” DRUP initially counted short-term stable disease (16 weeks) as a win. However, post-hoc analysis revealed that only half of those patients maintained that stability beyond 24 weeks. This distinction is vital; in the world of reimbursement and regulatory approval, “stable” is often a misleading proxy for “effective.”
The Forward Look: The Era of International Consortia
Looking ahead, the future of off-label drug repurposing cannot rely on national efforts alone. For rare cancers, the patient pool in a single country—even one as robust as the Netherlands—is simply too small to reach statistical significance before a drug’s patent expires.
We should expect to see a rapid shift toward the “consortium model” exemplified by PCM4EU and PRIME-ROSE. By aligning protocols and sharing data across European borders, researchers can accelerate enrollment and validate signals of activity while the drugs are still commercially viable. This international synergy is the only way to overcome the “patent cliff” that currently leaves patients stranded.
Additionally, we anticipate a tightening of clinical endpoints. The move away from “stable disease” toward more rigorous measures like Progression-Free Survival (PFS) of at least six months will become the gold standard. This will protect patients from ineffective therapies and ensure that only the most potent precision strategies move from experimental “off-label” use to national reimbursement.
Ultimately, the DRUP experience argues that the “right to try” must be balanced with the “duty to document.” Off-label precision medicine is not inherently ineffective, but it is too dangerous to be left to individual discretion. The path forward is a mandatory shift toward structured, data-generating frameworks.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.