Breakthrough CAR T-Cell Therapy Shows Promise for Generalized Myasthenia Gravis
In a landmark clinical trial, a novel CAR T-cell therapy engineered with mRNA technology has demonstrated significant efficacy in treating generalized myasthenia gravis (GMG). The findings, revealed today, offer a potential new avenue of hope for individuals battling this debilitating autoimmune disorder. A randomized, double-blind, placebo-controlled study revealed a substantially improved reduction in disease activity among patients receiving the innovative treatment compared to those given a placebo.
Understanding Generalized Myasthenia Gravis
Generalized myasthenia gravis is a chronic autoimmune neuromuscular disease that leads to fluctuating muscle weakness and fatigue. The condition occurs when the immune system mistakenly attacks the connections between nerves and muscles, disrupting communication and causing debilitating symptoms. These symptoms can include drooping eyelids, double vision, difficulty swallowing, and weakness in limb muscles. Current treatments often focus on managing symptoms, but a curative approach has remained elusive – until now.
How CAR T-Cell Therapy Works
Chimeric antigen receptor (CAR) T-cell therapy is a form of immunotherapy that harnesses the power of the patient’s own immune system to fight disease. In this specific application, T cells are extracted from the patient, genetically engineered to express a receptor (the CAR) that targets a specific protein, and then infused back into the patient. The engineered T cells then seek out and destroy cells expressing that protein. This particular therapy utilizes mRNA technology to engineer the CAR T-cells, offering a potentially more efficient and adaptable approach compared to traditional methods. The therapy targets BCMA, a protein found on certain immune cells involved in the autoimmune response in GMG.
The Clinical Trial: A Closer Look
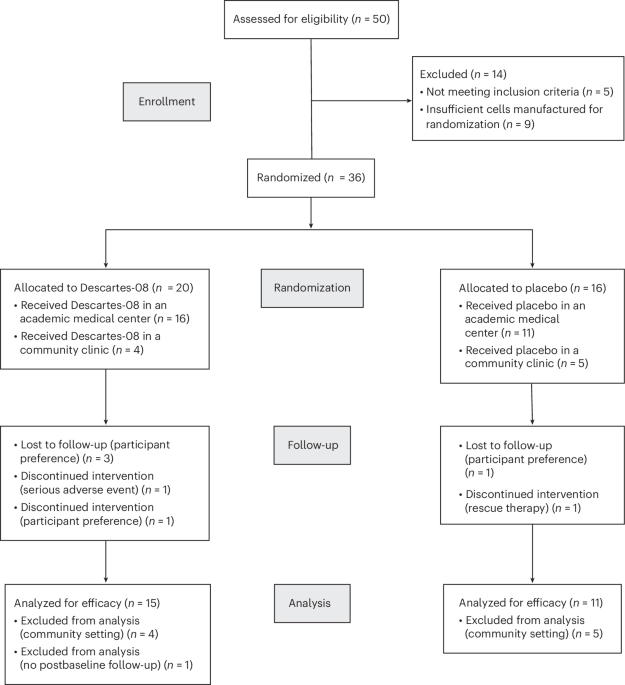
The clinical trial, detailed in Nature Medicine, involved a carefully selected cohort of patients with generalized myasthenia gravis. Participants were randomly assigned to receive either the autologous mRNA-engineered BCMA-targeting CAR T-cell therapy or a placebo. The study was designed as double-blind, meaning neither the patients nor the researchers knew who was receiving which treatment. The results showed a statistically significant and clinically meaningful reduction in disease activity in the treatment group. What does this mean for the future of GMG treatment? Could this approach be adapted for other autoimmune conditions?
Further research is underway to optimize the therapy, identify patients who are most likely to benefit, and assess the long-term durability of the response. The potential for a one-time treatment that could provide lasting remission from GMG is a game-changer for patients and their families.
For more information on autoimmune diseases and ongoing research, visit the Autoimmune Association.
Frequently Asked Questions About CAR T-Cell Therapy for Myasthenia Gravis
This groundbreaking research represents a significant step forward in the treatment of generalized myasthenia gravis. As the field continues to evolve, we can anticipate even more innovative therapies that offer hope and improved quality of life for those affected by this challenging condition.
Disclaimer: This article provides general information and should not be considered medical advice. Please consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
Share this article with your network to spread awareness about this exciting development! What are your thoughts on the potential of CAR T-cell therapy for autoimmune diseases? Join the discussion in the comments below.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.