Promising New Drug Combination Shows Benefit in Advanced Cancers
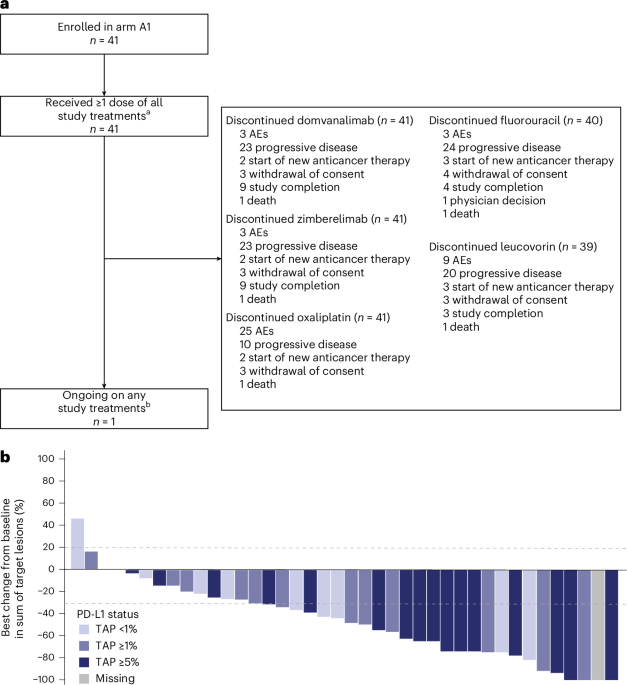
In a significant development for cancer treatment, a novel combination therapy is demonstrating encouraging results in patients battling advanced gastric, gastroesophageal junction, and esophageal cancers. The treatment regimen, featuring domvanalimab and zimberelimab alongside standard FOLFOX chemotherapy, was presented at the ESMO Congress 2025 and detailed in research published in Nature Medicine. Early data suggests improved response rates and survival outcomes, paving the way for a larger, phase 3 trial to confirm these findings.
Understanding the Treatment Approach
The combination therapy centers around two immunotherapy drugs: domvanalimab, an Fc-silent anti-TIGIT antibody, and zimberelimab, an anti-PD-1 antibody. These drugs work by unleashing the body’s own immune system to recognize and attack cancer cells. TIGIT and PD-1 are proteins that act as brakes on the immune system; blocking them can enhance the immune response. This immunotherapy approach is coupled with FOLFOX, a well-established chemotherapy regimen consisting of oxaliplatin, leucovorin, and fluorouracil. The synergy between these components appears to be driving the observed benefits.
The Landscape of Advanced Gastric and Esophageal Cancers
Advanced gastric and esophageal cancers remain challenging to treat, with limited therapeutic options available for many patients. These cancers often present at a late stage, making curative treatment difficult. Standard first-line treatment typically involves chemotherapy, but response rates are often suboptimal, and disease progression is common. Immunotherapy has emerged as a promising avenue for improving outcomes, but not all patients respond to these treatments. The current research explores a strategy to enhance the effectiveness of immunotherapy by combining it with chemotherapy and targeting multiple immune checkpoints.
The gastroesophageal junction is a particularly complex area, as cancers arising in this region can behave differently than those originating solely in the stomach or esophagus. This highlights the need for tailored treatment approaches and ongoing research to identify the most effective strategies for different subtypes of these cancers.
Did You Know?:
The phase 2 trial, conducted internationally, involved a single-arm design, meaning all patients received the combination therapy. While this design provides valuable preliminary data, it’s crucial to remember that the results are not directly comparable to those of a randomized controlled trial. The upcoming phase 3 trial will address this limitation by comparing the combination therapy to the current standard of care.
What role will biomarkers play in predicting which patients are most likely to benefit from this combination therapy? And how can we minimize the side effects associated with this intensive treatment regimen?
Frequently Asked Questions About This Cancer Treatment
The results from this phase 2 trial offer a glimmer of hope for patients facing these aggressive cancers. The upcoming phase 3 trial will be critical in determining whether this combination therapy can become a new standard of care.
Disclaimer: This article provides general information and should not be considered medical advice. Please consult with a qualified healthcare professional for any health concerns or before making any decisions related to your treatment.
Share this article with your network to spread awareness of this important research. Join the conversation in the comments below – what are your thoughts on the potential of immunotherapy combinations in cancer treatment?
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.