Promising New Vaccine Shows Safety and Immunity Against Typhoid and Salmonella
In a significant step forward in global health, a newly developed vaccine has demonstrated both safety and the ability to stimulate an immune response in healthy adults against typhoid fever and invasive non-typhoidal Salmonella infections. The findings, stemming from a rigorous phase 1 clinical trial, offer hope for a more effective preventative measure against these debilitating diseases, particularly in regions where they remain prevalent. This breakthrough could dramatically reduce the burden of illness and mortality associated with these bacterial infections.
Understanding Typhoid and Non-Typhoidal Salmonella Infections
Typhoid fever, caused by the bacterium Salmonella Typhi, is a life-threatening illness transmitted through contaminated food and water. Symptoms include high fever, headache, abdominal pain, and weakness. Invasive non-typhoidal Salmonella (iNTS) infections, caused by other Salmonella serovars, pose a particularly serious threat to individuals with weakened immune systems, especially in sub-Saharan Africa. Both diseases disproportionately affect children and individuals living in areas with poor sanitation and limited access to clean water. Current vaccines offer varying degrees of protection, and the development of a combined vaccine targeting both typhoid and iNTS represents a crucial advancement.
The Phase 1 Trial: A Stepwise Approach to Safety and Immunogenicity
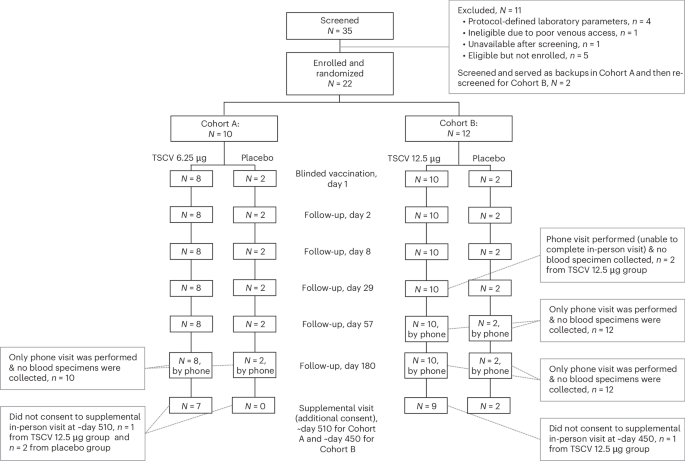
The recent research, detailed in Nature Medicine, employed a randomized, placebo-controlled, stepwise dose-escalation design. This method allows researchers to carefully assess the safety and immune response elicited by different doses of the conjugate vaccine. Healthy adult participants received escalating doses of the vaccine, while a control group received a placebo. Researchers meticulously monitored participants for adverse events and measured their immune response through blood tests. The results indicated that the vaccine was well-tolerated across all dose levels and consistently triggered a robust immune response.
What challenges remain in bringing this vaccine to widespread use, particularly in resource-limited settings? And how can global health organizations best collaborate to ensure equitable access to this potentially life-saving intervention?
Conjugate Vaccines: A Powerful Tool in Infectious Disease Prevention
The vaccine utilizes a conjugate technology, which enhances the immune response, particularly in young children. Conjugate vaccines link a bacterial surface antigen (a molecule that triggers an immune response) to a carrier protein. This conjugation process allows the immune system to recognize the antigen more effectively, leading to a stronger and more durable immune response. This technology has been successfully employed in vaccines against other bacterial diseases, such as Haemophilus influenzae type b (Hib) and pneumococcal disease.
Further research is now focused on evaluating the vaccine’s efficacy in larger, more diverse populations, including children. Phase 2 and 3 clinical trials are planned to assess the vaccine’s ability to protect against disease in real-world settings. The ultimate goal is to develop a vaccine that can be safely and effectively deployed to reduce the global burden of typhoid and iNTS infections.
For more information on typhoid fever and Salmonella infections, please visit the World Health Organization and the Centers for Disease Control and Prevention.
Frequently Asked Questions About the New Vaccine
- What is a conjugate vaccine and how does it work against typhoid and Salmonella? A conjugate vaccine links bacterial components to a carrier protein, boosting the immune response and providing stronger, longer-lasting protection.
- Was the vaccine tested on children during this phase 1 trial? No, this phase 1 trial focused on healthy adults. Further trials, including those involving children, are planned to assess the vaccine’s safety and efficacy in different age groups.
- How does this new vaccine differ from existing typhoid vaccines? This vaccine targets both typhoid fever and invasive non-typhoidal Salmonella infections, offering broader protection than many existing vaccines.
- What are the next steps in the development of this typhoid and Salmonella vaccine? Phase 2 and 3 clinical trials are planned to evaluate the vaccine’s efficacy in larger, more diverse populations in real-world settings.
- Where can I find more information about participating in clinical trials for this vaccine? Information about clinical trials can be found through research institutions and organizations involved in vaccine development, such as the National Institutes of Health.
- Is this vaccine expected to be widely available soon? While promising, the vaccine still requires further testing and regulatory approval before it can be widely distributed.
Share this article with your network to raise awareness about this important advancement in infectious disease prevention. Join the conversation in the comments below – what are your thoughts on the potential impact of this new vaccine?
Disclaimer: This article provides general information and should not be considered medical advice. Please consult with a healthcare professional for any health concerns or before making any decisions related to your health or treatment.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.