Breakthrough Zodasiran Trial Offers New Hope for Severe Cholesterol and Triglyceride Management
In a significant leap forward for cardiovascular medicine, new data reveals that zodasiran for cholesterol reduction is effectively slashing dangerous levels of blood fats in patients with genetic lipid disorders.
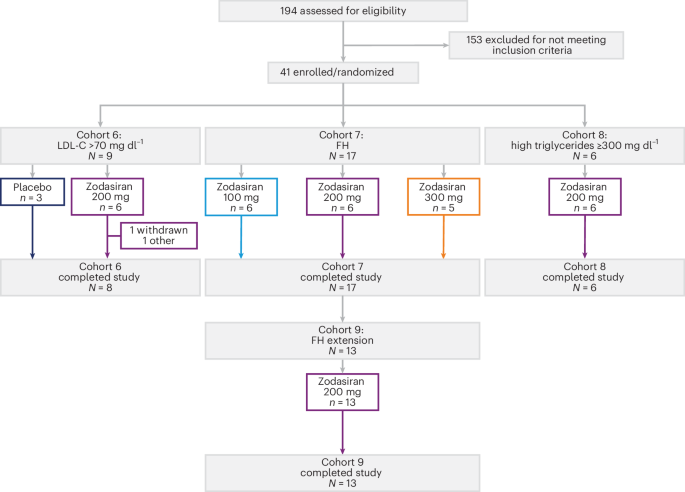
Results from a recent Phase 1 basket trial indicate that this experimental small interfering RNA (siRNA) therapy successfully targets the ANGPTL3 protein to provide dual-action relief for high-risk patients.
For those struggling with severe hypertriglyceridemia, zodasiran demonstrated a potent ability to drive down triglyceride levels. Even more promising were the results for patients battling heterozygous familial hypercholesterolemia (HeFH), where the drug lowered both triglycerides and low-density lipoprotein (LDL) cholesterol.
Cutting Through the Noise: How Zodasiran Targets ANGPTL3
The brilliance of zodasiran lies in its precision. Unlike traditional medications that attempt to manage symptoms, this siRNA therapy acts as a molecular “silencer.”
By targeting the ANGPTL3 gene, zodasiran prevents the body from producing a protein that typically inhibits the breakdown of fats. When this protein is suppressed, the body’s natural ability to clear LDL and triglycerides from the blood is unleashed.
This mechanism was detailed in a comprehensive study published in Nature Medicine, highlighting the drug’s efficiency across different patient profiles.
Understanding the Results: A Dual-Action Victory
The “basket trial” design allowed researchers to test the drug across multiple conditions simultaneously. The findings suggest that zodasiran is not a one-trick pony.
In patients with severe hypertriglyceridemia, the reduction in triglycerides is critical for preventing acute pancreatitis—a dangerous inflammation of the pancreas. Meanwhile, for the HeFH group, the combined drop in LDL and triglycerides addresses the root cause of accelerated atherosclerosis.
Could this shift the standard of care for genetic lipid disorders? Furthermore, how soon might we see these therapies move from clinical trials to pharmacy shelves?
The Science of siRNA and the Fight Against Cardiovascular Disease
To understand why zodasiran is a game-changer, one must look at the broader evolution of lipid management. For decades, statins have been the gold standard, yet a significant portion of the population remains “non-responders” due to genetic predispositions.
This is where the American Heart Association emphasizes the need for targeted therapies. Genetic conditions like HeFH mean that some individuals are born with a liver that cannot effectively remove LDL cholesterol from the blood, regardless of diet or exercise.
siRNA therapy represents a shift toward “precision medicine.” By silencing the proteins that block lipid clearance, scientists are essentially removing the brakes from the body’s own waste-disposal system for fats.
According to health guidelines from the Mayo Clinic, maintaining low LDL and triglyceride levels is the most effective way to reduce the risk of myocardial infarction and stroke. Zodasiran’s ability to tackle both simultaneously makes it a formidable tool in the preventative arsenal.
Frequently Asked Questions
What is zodasiran for cholesterol reduction?
Zodasiran is an experimental siRNA therapy that targets the ANGPTL3 protein to lower triglycerides and LDL cholesterol.
How does zodasiran for cholesterol reduction work?
It silences the production of ANGPTL3, allowing the body to clear fats from the bloodstream more efficiently.
Who is the ideal candidate for zodasiran for cholesterol reduction?
Patients with severe hypertriglyceridemia and heterozygous familial hypercholesterolemia (HeFH).
What were the primary results of zodasiran for cholesterol reduction in the trial?
The trial showed significant reductions in triglycerides for all patients and a dual reduction of LDL and triglycerides for those with HeFH.
Is zodasiran for cholesterol reduction a replacement for statins?
It targets a different pathway than statins and may provide an alternative for patients who do not respond to traditional therapy.
What phase of testing is zodasiran for cholesterol reduction currently in?
It has successfully completed a Phase 1 basket trial focusing on safety and initial efficacy.
Medical Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
Join the Conversation: Do you think genetic-silencing therapies are the future of heart health? Share this article with your network and let us know your thoughts in the comments below!
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.