New Hope for Spinal Muscular Atrophy: Phase 3 Trial Shows Promise of Intrathecal Gene Therapy
In a significant advancement for the treatment of spinal muscular atrophy (SMA), results from the Phase 3 STRENGTH study indicate that intrathecal onasemnogene abeparvovec demonstrates a favorable safety profile and offers a potential new therapeutic avenue for individuals affected by this debilitating genetic condition. The findings, released today, build upon previous successes with this gene therapy and suggest it could be a viable option even for patients who have previously received other treatments.
Understanding Spinal Muscular Atrophy
Spinal muscular atrophy is a rare, progressive neuromuscular disease that affects motor neurons – nerve cells in the spinal cord responsible for muscle control. Without these functioning neurons, muscles weaken and waste away, leading to increasing loss of mobility and, in severe cases, respiratory failure. The disease primarily affects infants and young children, though onset can occur at any age.
Currently, several treatments are available for SMA, including onasemnogene abeparvovec (Zolgensma), risdiplam (Evrysdi), and nusinersen (Spinraza). However, access to these therapies can be limited, and each has its own set of challenges, including administration methods and potential side effects. The STRENGTH study aims to expand the treatment landscape and provide more options for patients and their families.
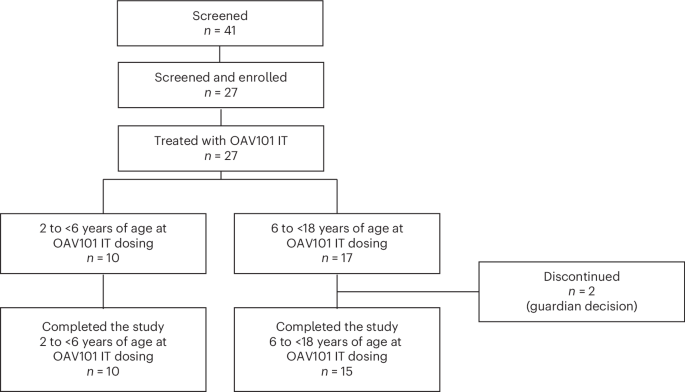
The STRENGTH Study: Key Findings
The STRENGTH study evaluated the safety and efficacy of intrathecal onasemnogene abeparvovec in a cohort of patients with SMA. Intrathecal administration, delivering the therapy directly into the cerebrospinal fluid, is a key aspect of this approach. The study’s results confirm a safety profile consistent with earlier trials involving treatment-naïve patients, meaning the observed side effects were manageable and predictable. This is particularly encouraging as it suggests the therapy can be safely administered to a broader range of individuals with SMA.
While detailed efficacy data is still being analyzed, the initial findings suggest a positive trend in motor function improvements among participants. Researchers are continuing to monitor patients for long-term outcomes. What does this mean for the future of SMA treatment? It suggests a move towards more personalized and accessible therapies.
The development of onasemnogene abeparvovec represents a landmark achievement in gene therapy. By delivering a functional copy of the SMN1 gene – the gene defective in SMA – the therapy aims to address the underlying cause of the disease, rather than simply managing its symptoms. But how will this new data impact current treatment protocols?
Expanding Treatment Options
The availability of multiple treatment options for SMA is a game-changer for patients and families. Each therapy has its own strengths and weaknesses, and the best approach will vary depending on individual circumstances, such as age, disease severity, and access to care. The STRENGTH study’s findings add another valuable tool to the arsenal of SMA treatments, potentially offering a more convenient and effective option for some patients.
Further research is needed to determine the optimal use of intrathecal onasemnogene abeparvovec and to compare its long-term outcomes to other available therapies. However, the initial results are undeniably promising and offer renewed hope for individuals living with spinal muscular atrophy.
Frequently Asked Questions About Spinal Muscular Atrophy and Gene Therapy
- What is intrathecal onasemnogene abeparvovec and how does it treat spinal muscular atrophy?
It’s a gene therapy that delivers a functional copy of the SMN1 gene directly into the cerebrospinal fluid, addressing the genetic root cause of SMA. - Is intrathecal onasemnogene abeparvovec safe for patients who have already received other SMA treatments?
The STRENGTH study suggests a favorable safety profile consistent with treatment-naïve patients, indicating it may be a viable option for those previously treated. - What are the potential benefits of intrathecal administration compared to other delivery methods?
Intrathecal delivery allows for direct access to the central nervous system, potentially maximizing therapeutic effect and minimizing systemic exposure. - How does the STRENGTH study contribute to our understanding of SMA treatment?
It provides further evidence supporting the safety and potential efficacy of intrathecal onasemnogene abeparvovec, expanding treatment options for SMA patients. - Where can I find more information about spinal muscular atrophy and available treatments?
Visit the Cure SMA website for comprehensive information, resources, and support. - What is the long-term outlook for individuals receiving intrathecal onasemnogene abeparvovec?
Long-term monitoring is ongoing to assess the durability of the therapeutic effect and identify any potential late-onset side effects.
The positive results from the STRENGTH study represent a significant step forward in the fight against spinal muscular atrophy. As research continues and new therapies emerge, the future looks brighter for individuals and families affected by this challenging condition.
Share this article to help raise awareness about SMA and the advancements being made in treatment! What are your thoughts on the potential of gene therapy for genetic diseases? Share your perspective in the comments below.
Disclaimer: This article provides general information and should not be considered medical advice. Please consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
Discover more from Archyworldys
Subscribe to get the latest posts sent to your email.